Phosphatidylinositol 5-phosphate (PI-5-P) is one of the seven phosphoinositides. They regulate a wide range of cellular functions. They localize in the nucleus, Golgi, endoplasmic reticulum, and at the plasma membrane. PI-5-P is an oxidative stress-induced regulator of AKT activation. The phosphorylation of the low abundance phosphoinositide PI-5-P at the 4-position, yielding the product phosphatidylinositol-4,5-bisphosphate (PI-4,5-P2), is catalyzed by PI5P4Kα, β, and γ. PI5P4K is important in several diseases. PI5P4Ks represent a lipid kinase family, and their underlying biology is important to numerous cellular processes. Therefore, the relevance of PI5P4K in a wide range of diseases has motivated efforts to develop PI5P4K inhibitors. In this study, THZ-P1-2 is a first-in-class and selective covalent PI5P4K inhibitor. The irreversible inhibition of PI5P4K by THZ-P1-2 compromises autophagy, an essential alternative energy source during periods of metabolic stress.
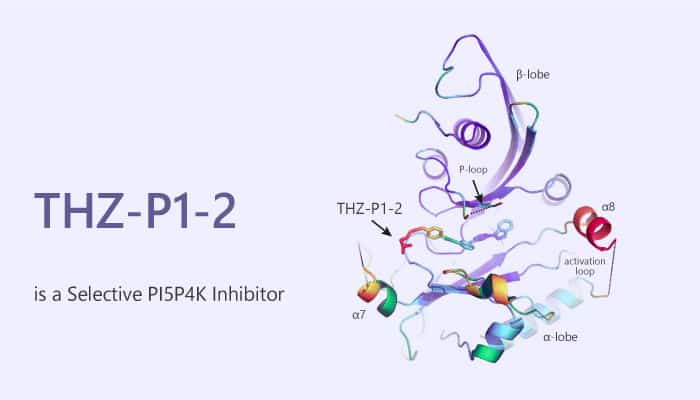
THZ-P1-2 is a first-in-class, and selective covalent PI5P4K inhibitor and causes autophagy disruption.
THZ-P1-2 covalently targets cysteines on a disordered loop in PI5P4Kα/β/γ. The IC50 is 190 nM for PI5P4Kα. THZ-P1-2 contains an electrophilic acrylamide moiety capable of undergoing a Michael addition. Thus, it binds in the active site and irreversibly inhibits the enzymatic activity of these kinases. Moreover, THZ-P1-2 exhibits approximately 75% inhibition of PI-4,5-P2 formation by PI5P4Kα and PI5P4Kγ and 50% inhibition by PI5P4Kβ at a concentration of 0.7 μM. In addition, THZ-P1-2 shows modest anti-proliferative activity in all six AML/ALL cell lines with IC50 values in the low micromolar range. THZ-P1-2 also phenocopies the effects of genetic deletion of PI5P4K α/β/γ in mice by causing autophagy defects in HeLa cells.
In summary, THZ-P1-2 is a first-in-class covalent inhibitor of the PI5P4K family. It exhibits durable cellular pharmacodynamics, causes autophagy dysfunction, and demonstrates modest anti-proliferative activity against leukemia-derived cell lines. All in all, PI5P4Ks are tractable targets, with THZ-P1-2 as a useful tool. It can further interrogate the therapeutic potential of PI5P4K inhibition and inform drug discovery campaigns for these lipid kinases in cancer metabolism and other autophagy-dependent disorders.
Reference: