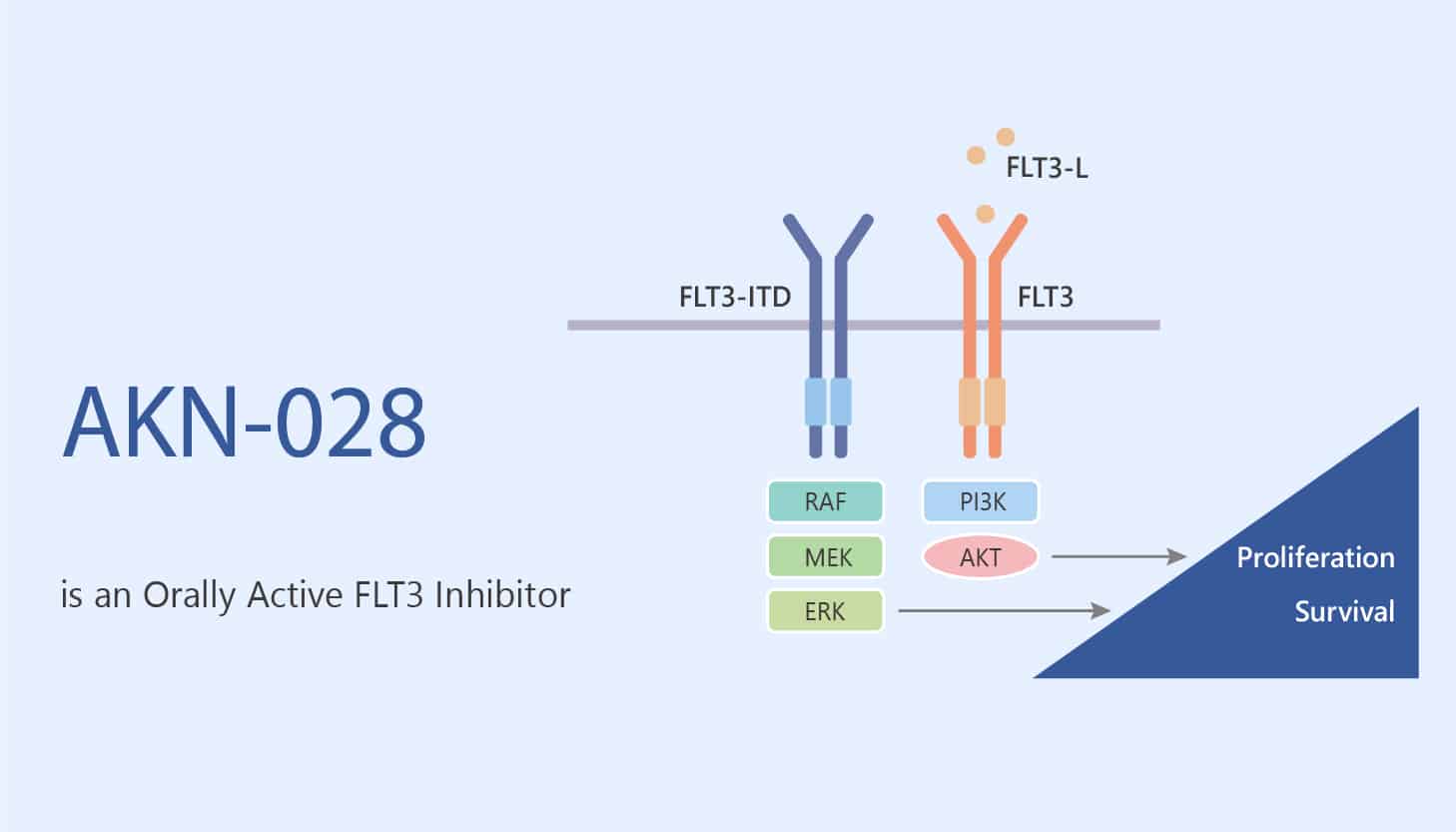
The activation of FMS like receptor tyrosine kinase 3 (FLT3) is a cytokine receptor that belongs to the receptor tyrosine kinase class III. Specifically, FLT3 signaling is important for the normal development of hematopoietic stem cells and progenitor cells. Besides, the FLT3 gene is one of the most common mutations in acute myeloid leukemia (AML). It is reported that some AML patients without FLT3 mutation have high levels of wild-type FLT3 in their embryonic cells. Moreover, FLT3 promotes cell survival and proliferation by interacting with multiple downstream targets. The targets include Akt, stat, and MAP kinase pathways. Furthermore, FLT3 is overexpressed in most AML patients. Therefore, the abnormally expressed receptor tyrosine kinase has become a promising target for the development of AML and other hematological malignancies. AKN-028 is an orally active and potent FLT3 tyrosine kinase inhibitor.
AKN-028 is an orally active and potent FLT3 tyrosine kinase inhibitor.
But, how does AKN-028 protect against AML via FLT3? Let’s discuss it in detail. First of all, AKN-028, a novel tyrosine kinase inhibitor (TKI), is a potent FMS-like receptor tyrosine kinase 3 (FLT3) inhibitor (IC50=6 nℳ). In addition, AKN-028 causes dose-dependent inhibition of FLT3 autophosphorylation. Meanwhile, AKN-028 triggers apoptosis in MV4-11 by activation of caspase 3. Nonetheless, AKN-028 (10 μM) is cytotoxic to AML cell lines and induces apoptosis in the AML cell line MV4-1. In the second place, AKN-028 exhibits anti-tumor activities for acute myeloid leukemia (AML) models with 15 mg/kg by subcutaneous injection twice daily. In mice, AKN-028 showed high oral bioavailability and anti-leukemia effect in primary AML and mv4-11 cells, and there is no major toxicity in the experiment. All in all, AKN-028 is an orally active and potent FLT3 tyrosine kinase inhibitor.
References:
A Eriksson, et al. Blood Cancer J. 2012 Aug 3;2(8):e81.