Acute myelogenous leukemia (AML) is a devastating hematologic cancer with limited treatments. Scientists have demonstrated that diverse genetic changes are associated with AML. Especially, the upregulation of tyrosine kinase signaling pathways stands for a common feature. Additionally, mutations in the Flt3 receptor tyrosine kinase accounts for almost one-third of AML cases. Multiple small-molecule Flt3 kinase inhibitors have entered into clinical trials for AML. Pan Flt3 inhibitors including midostaurin, lestaurtinib, and sorafenib, have limited efficacy. Compounds that inhibit common Flt3 resistance mutants are still lacked. A study from Mark C. Weir discovered and identified a novel inhibitor TL02-59 for Src-family kinase Fgr.
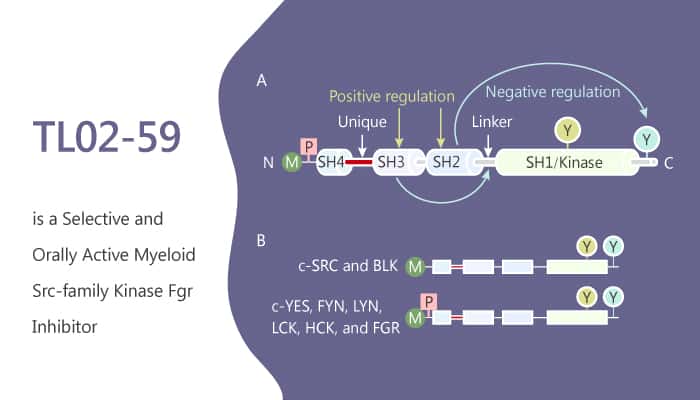
In vitro, TL02-59 inhibits the growth and induced apoptosis of AML cell lines expressing this kinase with single-digit nM potency. Besides, TL02-59 induces growth arrest in primary AML bone marrow samples. Moreover, TL02–59 selectively inhibits Fgr autophosphorylation in myeloid cells.
In vivo, TL02-59 has a t1/2 of 5.7 h by i.v injection and 6.5 h by p.o. administration, respectively. Additionally, TL02-59 (oral administration daily; 1 and 10 mg/kg; for three weeks) completely eliminate AML cells from the spleen and peripheral blood in a mouse model of AML, while dramatically suppressing bone marrow involvement.
Collectively, TL02-59 is an orally active, selective Src-family kinase Fgr inhibitor with an IC50 of 0.03 nM. TL02-59 also inhibits Lyn and Hck with IC50 values of 0.1 nM and 160 nM, respectively. TL02-59 potently suppresses acute myelogenous leukemia (AML) cell growth.