CBP (cyclic AMP response element-binding protein) and P300 (E1A binding protein) are two non-BET bromodomain oncology targets. Additionally, P300 is a paralogue of CBP, and they conduct similar activity. Specifically, CBP and P300 are multidomain proteins containing bromodomain and other domains, such as lysine acetyl transferase (KAT).
CBP/P300 bromodomains selectively recognize acetylated lysine residues. Recently, researchers also demonstrated that protein aggregation required CBP/P300 bromodomains and which disturbed proteostasis by impairing the ubiquitin proteasome system (UPS) and protein translation, leading to decreased cell viability. Moreover, the CBP/P300 bromodomains have been linked to disorders like acute myeloid leukemia (AML) and inflammatory and neurodegenerative diseases. Overall, CBP/EP300 are the most promising non-BET bromodomain targets for cancer and other pathological conditions.
In this article, we will introduce a potent and selective CBP/P300 bromodomain inhibitor, UMB298.
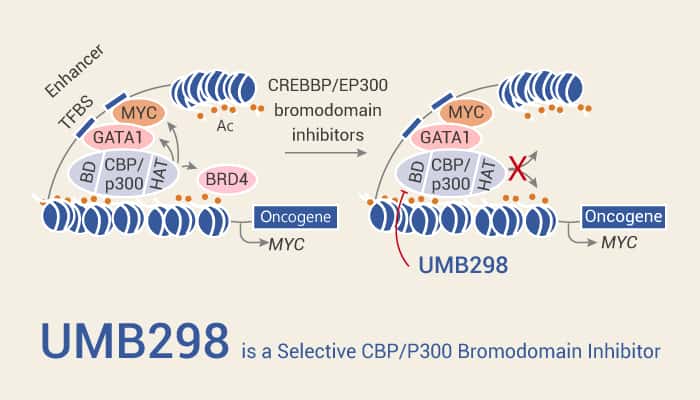
Generally, this compound has the potential to study acute myeloid leukemia. In addition, The IC50 values of UMB298 for CBP and BRD4 are 72 nM and 5193 nM, respectively. A research found that UMB298 inhibited MOLM13 cells growth. Moreover, UMB298 reduced the H3K27ac level similar to CBP30 and caused MYC depletion as a signature of CBP inhibition in acute myeloid leukemia. Certainly, the results suggest that UMB298 has excellent anti-cancer effect. Not only that, UMB298 displayed 72-fold selectivity for CBP over BRD4 as a selective CBP inhibitor. Consequently, these show that a central role for CBP/P300 bromodomain in anti-cancer models systems and the selectivity of UMB298.
In conclusion, UMB298 is a selective CBP/P300 bromodomain inhibitor. UMB298 inhibits cancer, could provide an effective study for acute myeloid leukemia.
References:
[1]. Muthengi A, et al. J Med Chem. 2021 May 13;64(9):5787-5801.