FLT3 is a kind of cytokine receptor, belonging to class III tyrosine kinase. Specifically, FLT3 signaling is important for the normal development of hematopoietic stem cells and progenitor cells. Specifically, the FLT3 gene is one of the most common mutations in acute myeloid leukemia (AML). There are high levels of wild-type FLT3 in embryo cells of AML patients without FLT3 mutation. Moreover, constitutively activated mutant FLT3 is a promising target for the treatment of the subgroup of patients with acute myeloid leukemia (AML) carrying it. Constitutively activated FLT3 is most commonly found in the form of internal tandem repeats (ITDs) in the near membrane domain 3. Moreover, transplantation of bone marrow cells infected with retrovirus expressing flt3-itd mutants can lead to the development of rapidly fatal myeloproliferative diseases in mice. ATH686 is a potent, selective, and ATP-competitive FLT3 inhibitor.
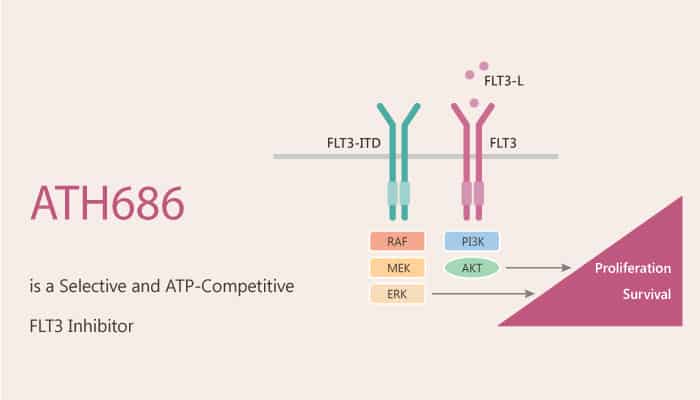
ATH686 is a potent, selective, and ATP-competitive FLT3 inhibitor with an antileukemic effect.
But, how does ATH68 protect against AML cells via FLT3? Let’s discuss it in detail. In the beginning, ATH686 is a potent, selective, and ATP-competitive FLT3 inhibitor antileukemic effect. Meanwhile, ATH686 targets mutant FLT3 protein kinase activity. Nonetheless, ATH686 inhibits the proliferation of cells harboring FLT3 mutants via induction of apoptosis and cell cycle inhibition.
In the second place, ATH686 potently inhibits cell proliferation (IC50 around 0.001 µM) via induction of apoptosis in FLT3-ITD-Ba/F3 cells and D835Y-Ba/F3 cells. Nonetheless, ATH686 inhibits autophosphorylation of mutant FLT3 in FLT3-ITD-Ba/F3 cells. Nonetheless, ATH686 selectively kills leukemia cells with mutant FLT3 by inhibiting FLT3 kinase activity but has no obvious effect on cells with wild-type FLT3.
All in all, ATH686 is a potent, selective, and ATP-competitive FLT3 inhibitor with an antileukemic effect.
References:
Ellen Weisberg, et al. Genes Cancer. 2010 Oct;1(10):1021-32.