Copper complexes play an important role in the active sites of a large number of metalloproteins in biological systems. Copper complexes also have a potential application for numerous catalytic processes in living organisms. In addition, copper complexes involve electron transfer reactions or activation of some antitumor substances. In this study, researchers reported the synthesis, characterization, and the minimal biologically active structural motif of soy Isoflavone, Genistein, and their copper(II) complex FPA-124.
Importantly, FPA-124 is a selective Akt inhibitor. Especially, FPA-124 shows 50% cell kill at concentrations of 7 µM in BT20 and 10 µM in PC-3 cells.
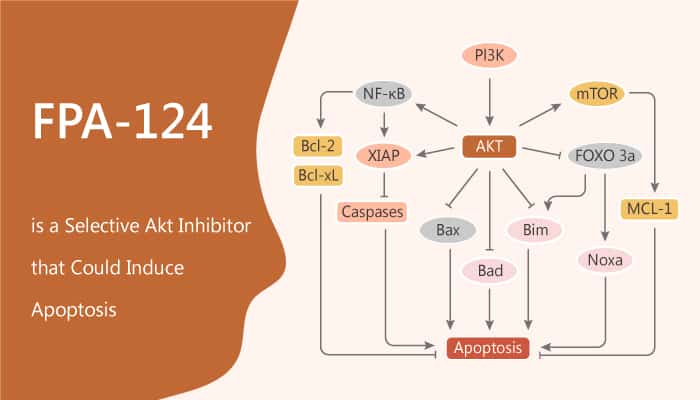
FPA-124 significantly forms stronger charge interactions in the kinase domain than Genistein, leading to better stabilization in the active pocket. Moreover, FPA-124 exhibits Akt protein inhibitory activities and causes NF-kB inactivation in a well-established orthotopic pancreatic tumor model using COLO 357 cells. Researchers also observed an inverse relationship between IC50 values of the anti-proliferative activities and the Cu2+/Cu+ redox couple for FPA-124 in multiple cancers. In particular, FPA-124 inhibits COLO357, BxPC3, BT20, and PC3 cancer cell lines proliferation with IC50 values of 34, 55, 7, and 10 μM in MTT cell proliferation assay. Furthermore, FPA-124 induces apoptosis in the entire cancer cell lines tested and FPA-124 shows the highest index of apoptotic cells in PC-3 cells.
In vivo, male 8-12-week-old C57BL/6J mice treated with FPA-124. Administered by gavage at the dosage of 20 mg/kg/bid every day. However, the combined treatment of Pioglitazone with FPA-124 has no effect on blood flow recovery evaluated by laser Doppler. All in all, FPA-124 is a cell-permeable inhibitor of Akt. Akt is an important regulator of growth and differentiation.