Caspases are cysteine proteases with strict specificity for cleaving peptide sequences C-terminal to aspartic acid residues. A prominent member of the pro-inflammatory class is caspase 1. It is responsible for the proteolytic activation of interleukin (IL)-1β and IL-18. IL-1β and IL-18 are cytokines. They play a major role in the immune response and numerous autoimmune and inflammatory diseases. Caspase 1 is constitutively and inducibly expressed in immune response elements such as T cells, macrophages, and neutrophils. Procaspase 1 associate with several multi-protein complexes capable of responding to numerous external stimuli. Therefore, caspase 1 is a major regulator of the inflammation response. Targeting proteases and specifically caspases, via small molecule therapeutics is an active area of research. Thus, inhibitors of caspase 1 have the potential for intervention strategies within ischemic disorders, Huntington’s disease, rheumatoid arthritis, osteoarthritis, inflammatory bowel disease, etc. ML132 is a potent and selective caspase 1 inhibitor.
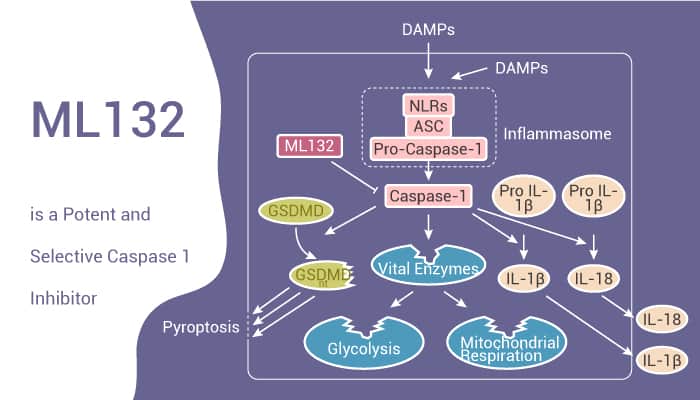
ML132 is a potent and selective caspase 1 inhibitor with an IC50 of 0.316 nM. To date, it is the most potent caspase 1 inhibitor. In addition, ML132 also possesses a unique selectivity pattern.
In summary, Caspases 1, also known as the interleukin-converting enzyme or ICE, is responsible for the proteolytic activation of interleukin (IL)-1β and IL-18. IL-1β and IL-18 are cytokines that play a major role in the immune response and within numerous autoimmune and inflammatory diseases. ML132 is a potent and selective caspase 1 inhibitor with an IC50 of 0.316 nM. Thus, inhibitors of caspase 1 have potential strategies within ischemic disorders, Huntington’s disease, amyotrophic lateral sclerosis (ALS), rheumatoid arthritis, osteoarthritis, inflammatory bowel disease, and sepsis.
Reference:
Boxer MB, et al. ChemMedChem. 2010 May 3;5(5):730-8.