Anaplastic lymphoma kinase (ALK) is a receptor tyrosine kinase and its mutation in domains. ALK Chromosomal translocations involve the kinase domain of ALK in many cancers.
In addition to ALCL, ALK fusion proteins are seen in diffuse large B-cell lymphoma (DLBCL), inflammatory myofibroblastic tumor (IMT), breast cancer, colorectal cancer, esophageal squamous cell cancer (ESCC), renal cell cancer (RCC), and non-small-cell lung cancer (NSCLC).
ALK fusion partners contribute to the dimerization of the ALK kinase domain. Besides, it increases autophosphorylation, thereby, causes the kinase to become constitutively active. There exist four kinase inhibitors for ALK-positive NSCLC treatment: crizotinib, ceritinib (LDK378), alectinib, and brigatinib. ALK-positive tumors display highly sensitive to ALK inhibition. However, the resistance to ALK inhibitors remains even though initial dramatic responses of variable median duration in inhibitors’ therapy.
The bivalent small molecules that enable to induce proteasome-mediated degradation of ALK is on the way. In this article, we will present an example of ALK PTOTAC degrader, TL13-112.
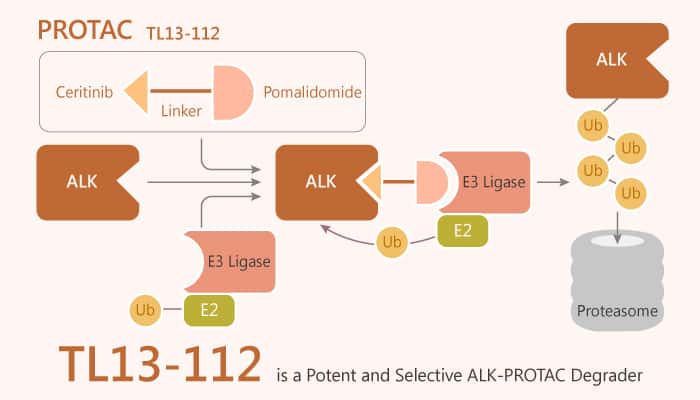
TL13-112 is a potent and selective ALK-PROTAC degrader and inhibits ALK activity an IC50 value of 0.14 nM. This PROTAC degrader also prompts the degradation of additional kinases including Aurora A, FER, PTK2, and RPS6KA1 with IC50 values of 8550 nM, 42.4 nM, 25.4 nM, and 677 nM, respectively.
Firstly, in Karpas 299 cells, TL13-112 induces a potent ALK degradation with a DC50 value of 40 nM. Also, it is selective for the degradation of ALK with the DC50s of 10 nM in H3122 cells.
ALK degradation acts at 4 hours of treatment in H3122 cells and 8 hours of treatment in Karpas 299 cells. And, the maximum degradation achieves at 16 hours in both cell lines.
TL13-112 also has the potential ability to overcome resistance mutation in Ba/F3 cells expressing EML4-ALK with the resistant mutations L1196M, C1156Y, or G1202R. This degrader shows a drop in anti-proliferative activity similar to the behavior of the parental ALK inhibitors. These results indicate that ALK targeting degraders based on ALK inhibitors enable to overcome resistance mutations and may show improved pharmacodynamic properties in these resistant mutant cell lines.
In conclusion, TL13-112 is a promising ALK degrader that performs good efficacy, we should attract more attention to its function in future investigation.
Reference:
Powell CE, et al. J Med Chem. 2018 May 10;61(9):4249-4255.