Deoxycytidine kinase (dCK) is an enzyme that is encoded by the DCK gene in humans. Specifically, dCK mainly phosphorylates deoxycytidine (DC) and converts DC into deoxycytidine monophosphate. Mall molecule dCK inhibitor can combine pharmacological disturbance of the de novo dNTP biosynthesis pathway. Besides, it can eliminate the acute lymphoblastic leukemia cells in animal models. The phosphorylation effect of dCK is to convert the deoxycytidine residue into the deoxycytidine monophosphate precursor (DCMP). Moreover, dCK also plays an important role in activating a variety of nucleoside analogs prodrugs widely used in anticancer and antiviral therapy. The kinase is relevant to the regulation of G2/M transformation in response to DNA damage in cancer cells. Furthermore, these aspects of dCK biology and its potential role as a new target for cancer treatment have led us to develop small molecular inhibitors with enzymatic activity. DI-82 is a potent deoxycytidine kinase (dCK) inhibitor.
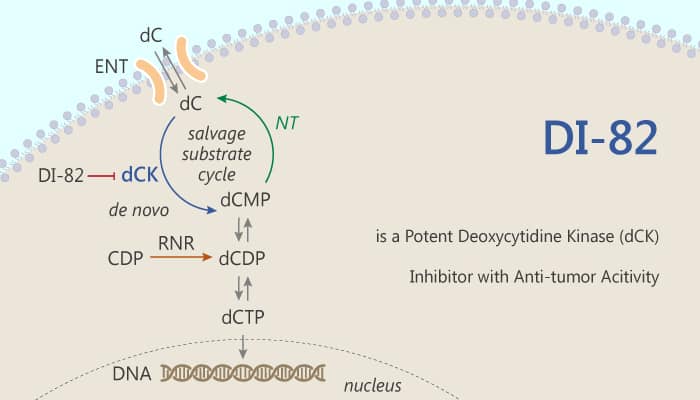
DI-82 is a potent deoxycytidine kinase (dCK) inhibitor with antitumor activity.
But, how does DI-82 protect against cancer cells via dCK? Let’s discuss it in detail. First of all, DI-82 is a potent dCK inhibitor with an IC50app of 27.8 nM and Kiapp of 9.2 nM. Meanwhile, DI-82 (compound 12R) has an IC50 of 3.7 nM in CCRF-CEM acute lymphoblastic leukemia cells. In the second place, DI-82 has an NADPH-dependent CLint of 22.7 μL/min•mg and an NADPH-dependent T1/2 of 102 mins in a standard microsomal liver clearance assay. Nonetheless, DI-82 completely blunts the ability of decitabine to bind human thymidylate synthase (TS). Last but not the least, DI-82 has significantly improved metabolic stability, making it a prime candidate for dCK-inhibitor-based therapies against hematological malignancies and, potentially, other cancers. All in all, DI-82 is a potent deoxycytidine kinase (dCK) inhibitor with antitumor activity.
References:
Julian Nomme, et al. J Med Chem. 2014 Nov 26;57(22):9480-94.