Focal adhesion tyrosine kinase (PTK2) is a cytoplasmic protein tyrosine kinase. PTK2 plays an important role in adhesion, spreading, motility, invasion, metastasis, survival, angiogenesis, epithelial to mesenchymal transition, cancer stem cells, and the tumor microenvironment. Overexpression and activation of PTK2 connect with several human malignant diseases, including colorectal, ovarian, esophageal, and hepatocellular carcinoma (HCC). HCC is the most common type of liver cancer in humans, accounting for 70−85% of primary liver malignancies, and is the third leading cause of cancer-related deaths worldwide. Several reports link PTK2 depletion or pharmacological inhibition of its kinase activity to the reduction of in vitro and in vivo tumorigenicity in HCC models. PROTACs represent an emerging therapeutic strategy to use small molecules to deplete a protein by repurposing the ubiquitin−proteasome system. In this study, BI-3663 is a highly selective PTK2/FAK PROTAC, with cereblon ligands to hijack E3 ligases for PTK2 degradation.
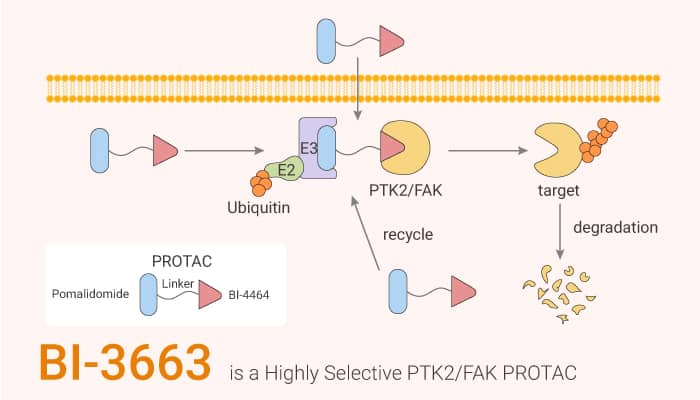
BI-3663 is a PROTAC that composes of BI-4464 linked to Pomalidomide with a linker. It also inhibits PTK2 (IC50=18 nM). Moreover, BI-3663 shows degradation potencies and efficacies in the 11 HCC cell lines (pDC50 = 7.45). Furthermore, it also potently degrades PTK2 in Hep3B2.1-7 cells, and A549 cells. The pDC50s are 7.6 and 7.9, respectively. All in all, BI-3663 shows anticancer activity.
In summary, BI-3663 is a highly selective E3 ligase-dependent PTK2 degrader (median DC50=30 nM). Furthermore, it shows overall comparable potencies and efficacies of PTK2 degradation in the 12 cell lines. In addition, BI-3663 has the potential for the research of hepatocellular carcinoma. It may provide valuable tools for the study of PTK2 degradation across different biological systems.
Reference:
Popow J, et al. J Med Chem. 2019 Mar 14;62(5):2508-2520.