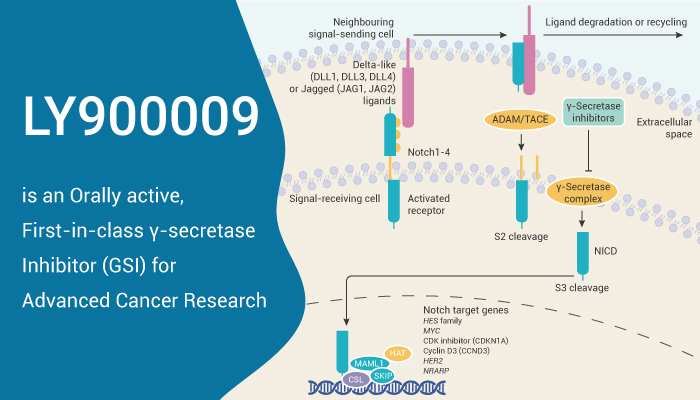
Gamma secretase is a multi-subunit protease complex, itself an integral membrane protein, that cleaves single-pass transmembrane proteins at residues within the transmembrane domain. Importantly, Gamma secretase is an internal protease that cleaves within the membrane-spanning domain of its substrate proteins, including amyloid precursor protein (APP) and Notch. Mainly, Notch signaling regulates stem cell development and survival and is deregulated in multiple malignancies. Specifically, Notch signaling regulates the expression of receptor tyrosine kinases, including the pro-angiogenic vascular endothelial growth factor receptor-1 and the pro-growth epidermal growth factor receptor. Moreover, Evidence suggests that deregulated Notch 1-4 signaling is also implicated in numerous malignancies. Also, Notch signaling is a component of stem cell fate and differentiation in various tissues. Therefore, inhibition of Notch signaling via γ-secretase may be an attractive cancer therapy target.
In this article, we will introduce a potent, selective, and orally active γ-secretase Inhibitor, LY900009.
LY900009 is a γ-secretase protein and inhibits the cleavage of NICD. Besides, LY900009 inhibits Notch signaling in tumor cell lines and endothelial cells with IC50 values range 0.005-20 nM. Meanwhile, LY900009 (3 mg/kg; p.o.; once) dose-dependently inhibits Notch cleavage and induces apoptosis after 24 h in a xenograft tumor model. At the same time, LY900009 reveals inhibition of angiogenesis through the formation of leaky vasculature and produces tumor regression in Notch-dependent tumor models in a rat model. Furthermore, LY900009 had tumor regression in Notch-dependent tumor models. Other, Studies show that LY900009 inhibits plasma levels of amyloid-β peptide in a dose-dependent manner with 80-90% inhibition.
All in all, LY900009 is a potent, selective, and orally active inhibitor of Notch signaling via selective inhibition of the γ-secretase protein. Additionally, LY900009 has the potential for research on advanced cancer.
Reference:
[1] Pant S, et al. Eur J Cancer. 2016 Mar;56:1-9.