Matrix metalloproteinases (MMPs) are a family of zinc- and calcium-dependent endopeptidases. They are responsible for the degradation of most extracellular matrix proteins during organogenesis, growth and normal tissue turnover. Elevated MMP expression and activity has existed in numerous disease processes, including inflammation, tumor cell metastasis, and atherosclerosis.
At present, synthetic MMP inhibitors available for the treatment of degenerative diseases are hydroxamate based pseudopeptides and nonpeptidyl sulfonamide-based hydroxamic acid inhibitors. Beside this therapeutical application, MMPIs are also potentially useful for the noninvasive in vivo imaging of activated MMPs. Therefore, radiolabeled analogues of MMPIs can serve as radiotracers for the detection of unbalanced MMP levels. Such improved diagnosis of diseases associated with dysregulated MMP activity would be of great clinical interest and strongly influence therapy and diagnosis algorithms.
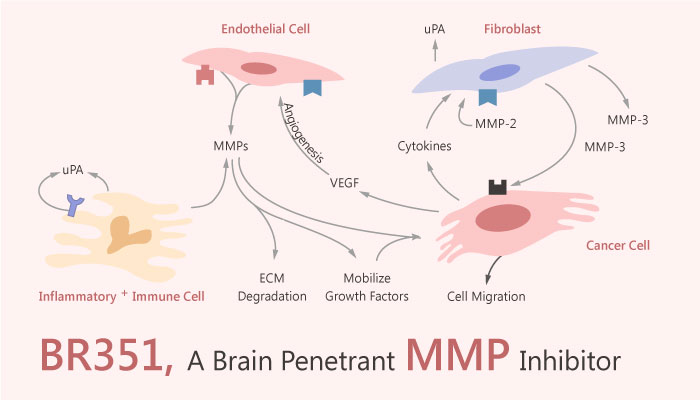
Targeting MMP with PET activity-based imaging probes could offer a non-invasive tool to early detect and diagnose MMP-related diseases. However, Most of them are broad spectrum MMP inhibitors and currently there are no successful MMP selective radiotracers available. BR351 is also a brain penetrant broad spectrum MMP PET radiotracers with favourable affinities for activated MMP-2 and MMP-9. It is a potential tools for the molecular imaging of activated MMPs with PET.
Vazquez N et al evaluated [18 F]BR351 as potential MMP-9 PET radiotracers in a colorectal cancer model expressing MMP-9. [18 F]BR351 was quickly metabolized and displayed only low tumor uptake. Hence, these unpromising results make this compound not suited for MMP imaging in mouse models of cancer.
Reference:
J Labelled Comp Radiopharm. 2017 Jan;60(1):69-79.