The ubiquitin proteasome system participates in a variety of biological functions including cell cycle progression, intracellular signaling and protein degradation. Therefore, it is not surprising to find many misregulation of this system in cancer. Fortunately, the first-generation deubiquitylating enzyme (DUB) inhibitors are now approaching clinical trials. For instance, the clinical success of Bortezomib for treatment of multiple myeloma proves that targeting the ubiquitin proteasome system is valid. However, the clinical development of selective DUB inhibitors has been challenging. In a book edited by By Nishi Kumari, the authors discussed the prominent issues and highlighted recent advances.
By regulating ubiquitin homeostasis, DUBs have been implicated in tumorigenesis. Hence, it is common that deregulation of DUBs may result in severe pathological conditions. A number of inhibitors including both specific and pan-enzyme inhibitors for DUB have been identified. Additionally, they are undergoing been explored for its affection in patients. As a result, DUBs show a great degree of substrate specificity. DUBs also have an active site such as the catalytic cysteine which makes DUBs attractive targets for small molecule drug discovery. A majority of the DUBs inhibitors are capable of forming covalent adducts which in turn blocks the DUBs activity. A diverse number of compounds for DUBs have been identified and studied.
In the review, the authors remained a novel deubiquitylating enzyme (DUB) inhibitor, GSK2643943A. It targets USP20/Ub-Rho and shows an IC50 of 160 nM.
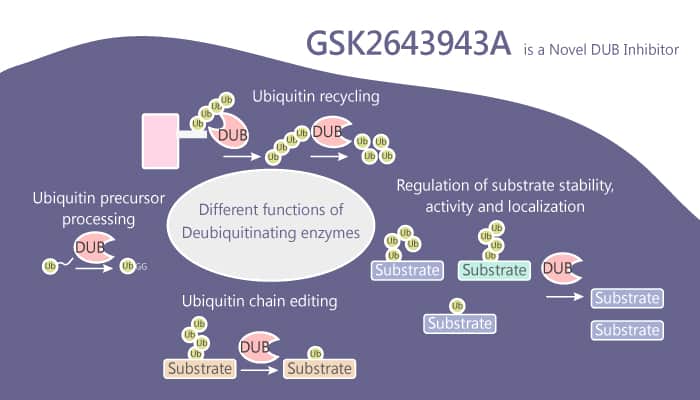
And the scientists have a lot to do for the lacked further study.
Nishi Kumari, et al. Targeting the Ubiquitin Proteasome System in Cancer. Neoplasm. 2017 Dec.