The three human RAS genes [KRAS, NRAS and HRAS] encode four RAS proteins, with two KRAS isoforms. The two KRAS isoforms arise from alternative RNA splicing KRAS4A and KRAS4B. The 3 human RAS genes belong to the protein family of small GTPases. GTPases function as binary molecular switches involved in cell signaling. In particular, RAS proteins (KRAS4A, KRAS4B, NRAS and HRAS) function as GDP-GTP-regulated binary on-off switches, which regulate cytoplasmic signaling networks that control diverse normal cellular processes. KRAS is the most frequently mutated oncogene, with mutation rates of 86-96% in pancreatic cancers, 40-54% in colorectal cancers, and 27-39% in lung adenocarcinomas. NRAS is predominantly mutated in melanoma and hematological malignancies, while HRAS mutations are salivary gland and urinary tract cancers.
BI-2852 is a KRAS inhibitor that binds with nanomolar affinity to a pocket, between switch I and II on RAS. Especially, BI-2852 is mechanistically distinct from covalent KRASG12C inhibitors because BI-2852 binds to a different pocket present in both the active and inactive forms of KRAS. Furthermore, BI-2852 modulates pERK and pAKT and has an antiproliferative effect in KRAS mutant cells. In cells, BI-2852 inhibits SOS1-catalyzed exchange of GDP to GTP as well as GAP-catalyzed exchange of GTP to GDP. This effect results in no net change in cellular GTP-RAS levels upon treatment. Moreover, BI-2852 reduces pERK and pAKT levels in a dose-dependent manner. This effect leads to an anti-proliferative effect in NCI-H358 cells.
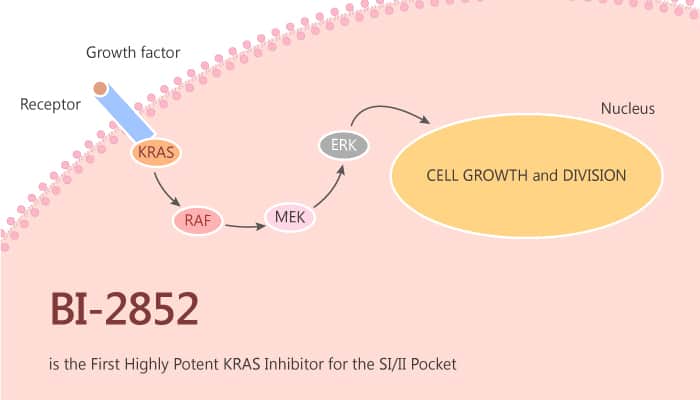
To summarise, BI-2852 will also serve as a useful chemical probe for the scientific community in the study of RAS biology of simultaneous inhibition of active and inactive RAS in an in vitro setting.