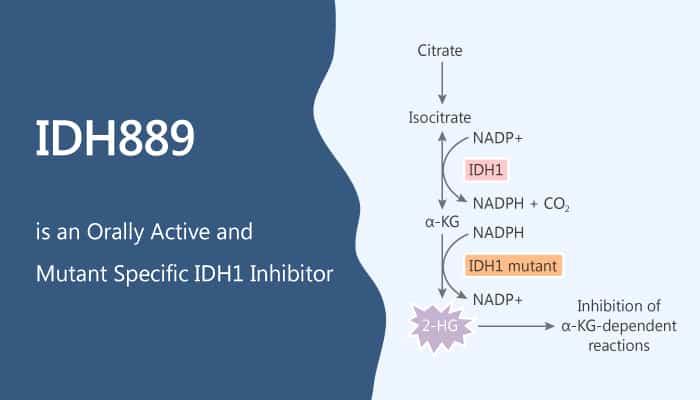
Heterozygous mutations in human cytoplasmic isocitrate dehydrogenase 1 (IDH1) at Arg132 (R132) occur in multiple cancer types, including acute myeloid leukemia, glioma, chondrosarcoma, and cholangiocarcinoma. In this study, Julian R. Levell, et al identified IDH889, which is a potent inhibitor of IDH1R132H by high throughput screening. Especially, IDH889 binds into an allosteric, induced-fit pocket in IDH1R132H. Moreover, IDH889 has good overall selectivity vs the wild-type protein, and inhibits both the IDH1R132H and IDH1R132C mutants. Furthermore, IDH889 has broad utility across the various known R132 mutations. IDH889 also has excellent permeability and no efflux in the Caco-2 and human MDR1-MDCK cell lines.
IDH889 can penetrate the blood-brain barrier. To assess brain/plasma ratio of IDH889 in a rat pharmacokinetics study, researchers demonstrate favorable distribution to the brain of IDH889. The result is 30 mg/kg po with the 1.4 brain/blood ratio. IDH889 also demonstrates significantly improved plasma exposure in mice vs IDH662 (AUC 3.6 μM•h, Cmax 1.7 μM at 10 mg/kg; AUC 55.5 μM•h, Cmax 14.2 μM at 100 mg/kg). To illustrate, researchers optimized the high throughput screening hit for potency and PK properties to generate IDH889 suitable for exploring the effect of inhibition of production of 2-HG by IDH1R132H in vitro and in vivo cancer models. Oral dosing of IDH889 in a murine IDH1 mutant tumor xenograft model shows robust reduction of tumor derived 2-HG, a PD biomarker of mutant-IDH1R132 activity.
In conclusion, IDH889 is a potent inhibitor of IDH1R132 for treating patients with IDH1 mutant brain cancers.