Choline kinase (CHK) catalyzes the phosphorylation of choline by ATP in the presence of Mg2+, yielding phosphocholine. This enzyme step commits choline to the Kennedy pathway for the biosynthesis of phosphatidylcholine, the major phospholipid constituent in biomembranes. Phosphatidylcholine has an essential structural function as well as serving as a reservoir for lipid second messengers. Furthermore, there are two enzymes in mammals: CHKα and CHKβ. CHKβ has not been reported to be oncogenic, but CHKα participates in initiation and progression of several tumors. In this study, CHKα plays a crucial role in the regulation of membrane phospholipid synthesis, and has oncogenic properties in vitro. However, pancreatic ductal adenocarcinoma (PDAC) increases CHKα expression. MN58b, a first-generation choline kinase inhibitors (CHKI)-competitive inhibitor, has antiproliferative and antitumoral activity in vitro and in vivo.
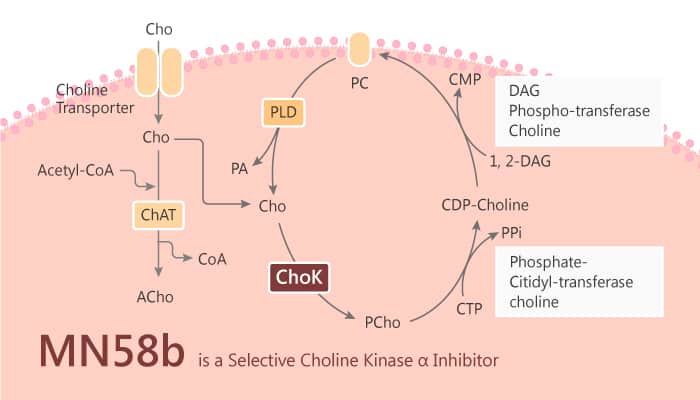
MN58b selectively inhibits CHKα. Accordingly, there is a decrease in the synthesis of phosphocholine from choline in IMIM-PC-2 cells. MN58b has a marked effect on colony formation at 1 μM, and it completely abolishes SK-PC-1, Suit2 008, IMIM-PC-2, and RWP-1 cells growth at 5 μM. In a panel of 12 PDAC cell lines, the IC50 of MN58b ranges from 0.23 to 3.2 μM. In addition, MN58b induces apoptosis and this response correlates with CHKα expression. MN58b with other chemotherapeutic agents is active in PDAC therapy.
MN58b treatment significantly decreases phosphomonoesters in both HT29 and MDA-MB-231 xenografts.
All in all, MN58b is a selective CHKα inhibitor. It results in inhibition of phosphocholine synthesis. MN58b reduces cell growth through the induction of apoptosis, and also has antitumoral activity. Overall, CHKα inhibition merits further attention as a therapeutic option in patients with PDAC and that expression levels may predict response.
Reference:
Mazarico JM, et al. Mol Cancer Ther. 2016 Feb;15(2):323-33.
Al-Saffar NM, et al. Cancer Res. 2006 Jan 1;66(1):427-34.