Pancreatic cancer is the fourth leading cause of cancer-related deaths in Japan. Gemcitabine has been the standard therapy for advanced pancreatic cancer for over a decade. Additional novel combination therapies have been developed. It includes gemcitabine plus erlotinib; a combination of leucovorin, 5-fluorouracil, irinotecan, and oxaliplatin; and gemcitabine plus nab-paclitaxel. However, the effect remains insufficient, and it is, therefore, necessary to develop a novel effective strategy for pancreatic cancer treatment. PPARγ belongs to the nuclear hormone receptor transcription factor superfamily. They play major roles in adipogenesis, glucose metabolism, and angiogenesis. A variety of normal tissues and tumors (such as pancreatic cancer) express PPARγ. The thiazolidinedione family of PPARγ agonists, such as Troglitazone (TGZ), pioglitazone (PGZ), and rosiglitazone, regulate growth and survival in a number of cancer cell lines. Thus, Troglitazone may serve as an effective approach for the treatment of pancreatic cancer.
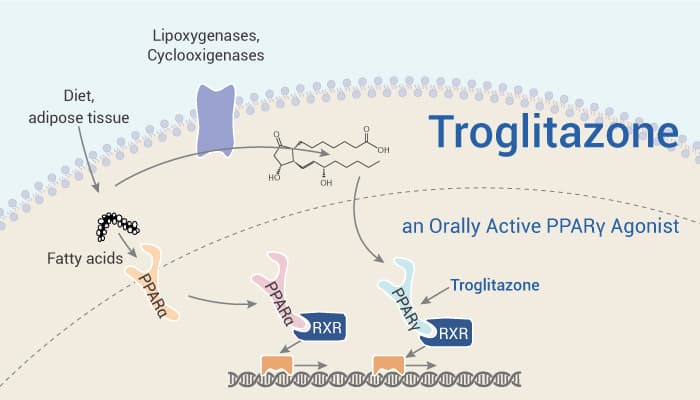
Troglitazone (TGZ) is a peroxisome proliferator-activated receptor-gamma (PPARγ) agonist. It has EC50s of 550 nM and 780 nM for human and murine PPARγ receptor, respectively. In addition, Troglitazone shows dose-dependent cytotoxicity. The IC50 values of 49.9 and 51.3 μM in MIA Paca2 and PANC-1 cells, respectively. Troglitazone also significantly increases the activity of caspase-3 by approximately 3.5-fold in MIA Paca2 cells. Moreover, Troglitazone markedly decreases Bcl-2 expression by approximately 70% in MIA Paca2 cells and moderately decreases levels in PANC-1 cells. Furthermore, Troglitazone exhibits inhibitory effects on tumor growth in the MIA Paca2 xenograft model. TGZ also exhibits inhibitory effects on tumor growth in the MIA Paca2 xenograft model.
In summary, Troglitazone induces mitochondria-mediated apoptosis in MIA Paca2 cells. Troglitazone is a potent PPARγ agonist and is a potential chemopreventive and chemotherapeutic agent.
Reference:
Fujita M, et al. J Exp Clin Cancer Res. 2017;36(1):91. Published 2017 Jul 3.