KRAS (Kirsten rat sarcoma virus) is one of the most commonly altered oncogenes in human cancers. Recurrent oncogenic mutations in KRAS can reprogram glutamine metabolism to increase the production of NADPH to control ROS. Besides, mutant KRAS promotes the expression of nucleotide synthesis genes and controls glucose uptake and flux through the PPP through incompletely understood mechanisms. KRAS can provide instructions for making a protein called K-Ras, a part of the RAS/MAPK pathway. What’s more, the K-Ras protein is a GTPase, a class of enzymes that convert the nucleotide guanosine triphosphate (GTP) into guanosine diphosphate (GDP). The protein relays signals from outside the cell to the cell’s nucleus. These signals instruct the cell to grow and divide or to mature and take on specialized functions.
KRAS G12S is a specific variation in the KRAS protein. KRAS with no mutation at amino acid position 12 has a glycine, or G for short. The amino acid at position 12 in KRAS with the G12S mutation is a serine, or S for short. Importantly, K-Ras G12S is an oncogenic driver with GTPase activity. The KRAS p.G12S mutation has been observed in thousands of patient tumors36, occurring in 2.8% of colorectal adenocarcinoma and 2.5% of nonsmall cell lung cancer.
G12Si-5 is a potent and reversible inhibitor of K-Ras G12S.
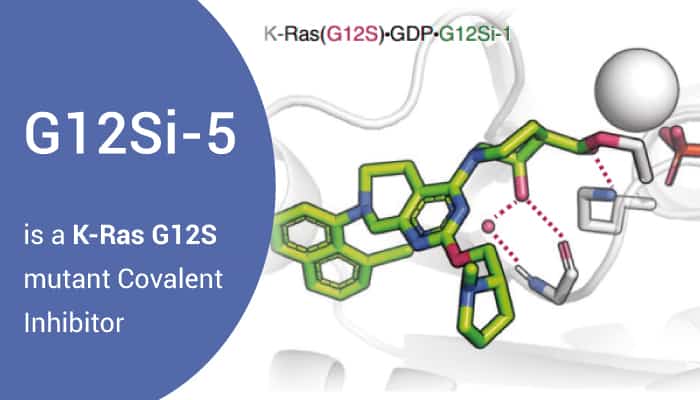
G12Si-5 is a potent, selective and reversible covalent inhibitor of the G12S mutant of K-Ras. Specifically, G12Si-5 binds with the S-IIP domain and suppresses oncogenic signaling. Inhibition of Ras signaling by G12Si-5 in A549 cells was dose-dependent. Therefore, G12Si-5 led to nearly complete loss of Ras•GTP and concomitant inhibition of phospho-ERK in A549 cells. Meanwhile, G12Si-5 did not perturb the signaling in A375 cells or SW1990 cells, and showed only weak inhibition of phospho-Erk in H358 cells, an effect we ascribed to possible ring opening of the β-lactone by the nucleophilic cysteine.
All in all, G12Si-5 is a potent, selective, and reversible covalent inhibitor of the K-Ras G12S.
Reference:
[1]. Zhang Z, et, al. Nat Chem Biol. 2022 Jul 21.