Double-strand break repair protein MRE11 is an enzyme that in humans is encoded by the MRE11 gene. The MRE11/Rad50/NBS1 (MRN) complex maintains genomic stability by bridging DNA ends, and initiating DNA damage signaling through activation of the ATM kinase. Mre11 possesses DNA nuclease activities in evolution, but play unknown roles in mammals. Importantly, MRE11 nuclease forms the core of the MRE11-RAD50-NBS1 (MRN) complex, which has essential roles in detecting, signaling, protecting and repairing DNA double strand breaks (DSBs). In prokaryotes, the MRE11 protein interacts with the Rad50 protein. Appears to have an active role in repairing DNA damage experimentally introduced by γ-radiation. However, the endo- and exonuclease activities of MRE11 regulate DNA double-strand break repair by nonhomologous end-joining (NHEJ) versus homologous recombination (HR) remains enigmatic.
PFM39, a Mirin analog, is a potent and selective MRE11 exonuclease inhibitor. At the same time, PFM39 does not inhibit TmMre11 or human MRN endonuclease activity on ssDNA circles. Indeed, structures suggested that, PFM39 has an amino group in place of the hydroxyl group, bound in the pocket between the His61 loop and the adjacent loop (Asn93-Lys97). In addition, in cells with chromosomally integrated I-SceI inducible DSBs, PFM39 inhibits homologous recombination (HR) without significantly increasing NHEJ. Moreover, PFM39 relieves the DNA double-strand break repair (DSBR) defect caused by EXO1/BLM depletion in EXO1/BLM depleted cells. At the same time, PFM39 inhibits phosphate rotation for dsDNA exonuclease activity.
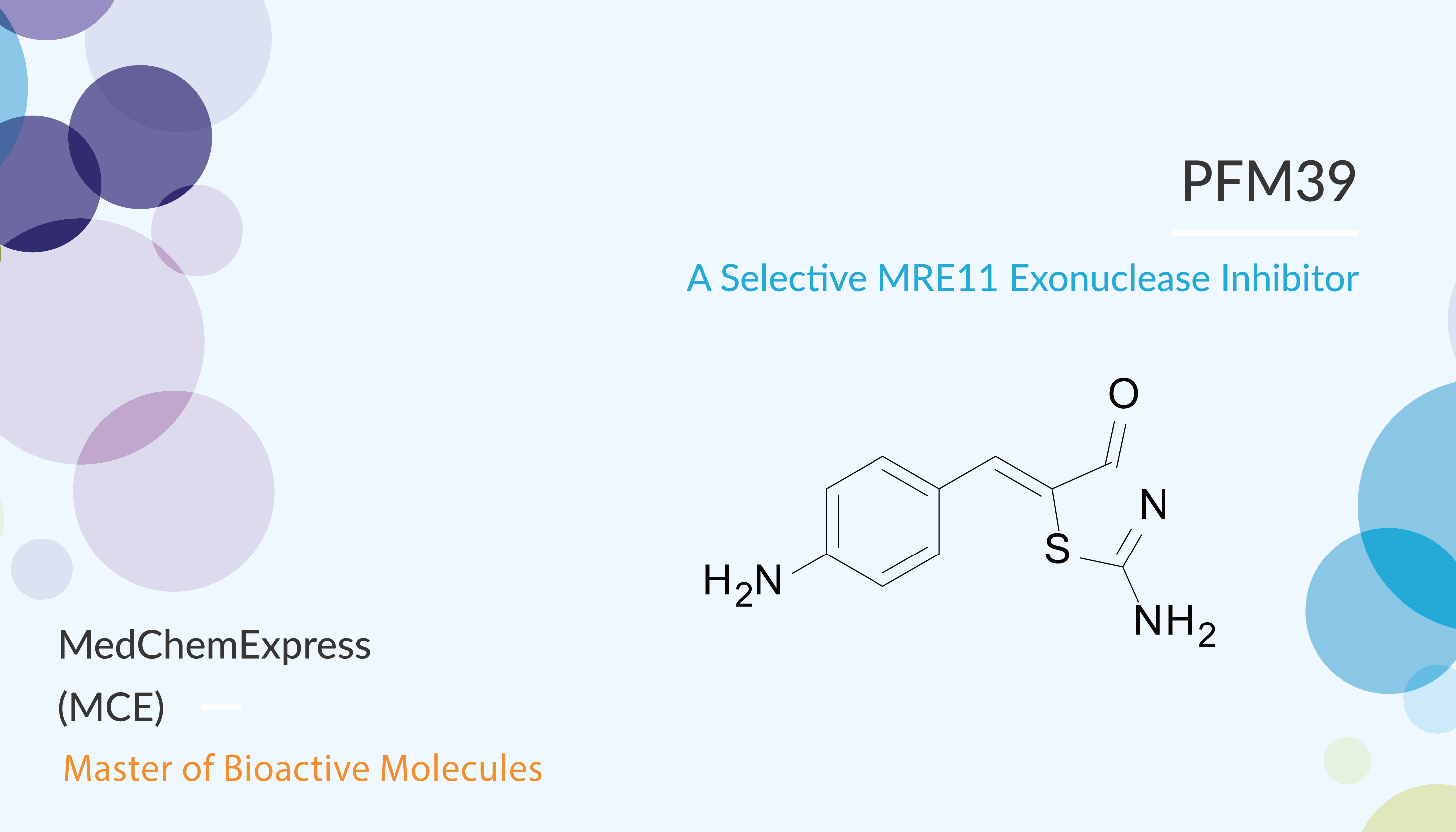
To sum up, PFM39 is a potent and selective MRE11 exonuclease inhibitor, and does not inhibit endonuclease, nuclease activity.
References:
[1] Atsushi Shibata, et al. Mol Cell. 2014 Jan 9;53(1):7-18.