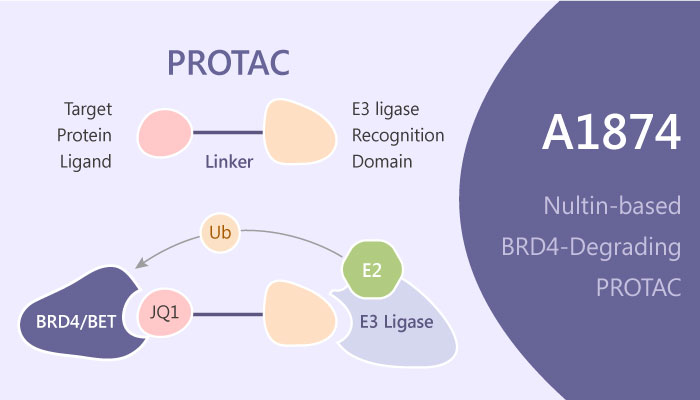
In recent years, the studies associated with PROTAC have grown steadily. And Although the E3 ligase MDM2 is bound by the nutlin class of small-molecule ligands, there are few nutlin-based PROTAC. Because a nutlin-based PROTAC should both downregulate its target protein and upregulate p53. A study from John Hines et al discovered and identified a novel nultin-based BRD4-degrading PROTAC, A1874.
This compound represented the first BRD4-targeting MDM2-based PROTAC that possesses potent biological activities in inhibiting cancer cells proliferation.
In vitro, A1874 showed a DC50 of 32 nM (induce BRD4 degradation in cells). Furthermore, treatment of HCT116 cells of A1874 (0-10 μM, 24 hours) induced a dose-dependent knockdown of BRD4 levels, with near-maximum knockdown by 100 nM and a maximum degradation (Dmax) of BRD4 of 98% of the levels in control (0.1% DMSO-treated) cells. In addition, treatment of HCT116 cells of A1874 (0-10 μM, 24 hours) increased p53 levels in the HCT116 cells and showed dose-dependent p53 stabilization. That was to say, A1874 represented a much improved nutlin-based protein degrader, approaching the activity of other reported PROTACs.
To further investigate the impact on down signaling induced by A1874, the authors measured the express of c-Myc. Actually, A1874 treatment significantly reduced c-Myc expression by 85% compared to control HCT116 cells, which is more potent than JQ1. What’s more, A1874 showed dose-dependent p53 stabilization by increasing p53 levels in the HCT116 cells by immunoblotting. Notably, the concentration of 250 nM of A1874 significantly induced p21CIP1/WAF1 and only caused small increases in p53 levels, reflecting the amplification associated as a signal is transduced downstream.
However, more in vivo experiments need to be carried out to verify the function of A1874 in animal models. We’re looking forward to see the promising result.
Reference:
Hines J, et al. Cancer Res. 2019 Jan 1;79(1):251-262.