Ubiquitin Proteasome System
The ubiquitin proteasome system (UPS) consists of two separate, consecutive steps: ubiquitylation and proteasomal degradation. In particular, the ubiquitin proteasome system is the principal pathway for the clearance of short-lived, damaged, and mis-folded proteins in the nucleus and cytoplasm. The ubiquitin proteasome system controls the degradation of intracellular proteins. Gratifyingly,targeting protein degradation is a reasonable strategy for cancer therapy. Consequently, the ubiquitin-proteasome system is responsible for the regulated degradation of intracellular proteins with important roles in a broad array of cellular functions.
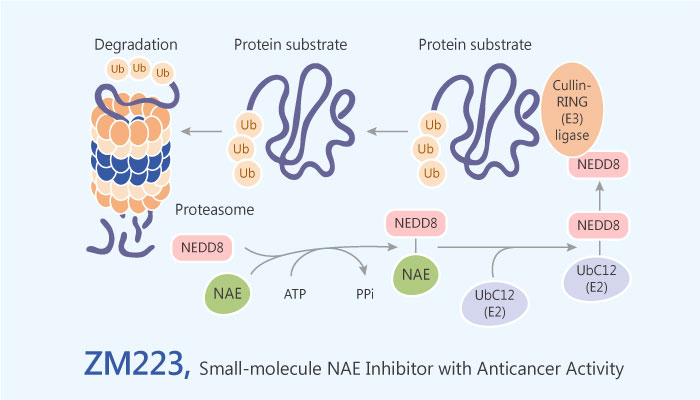
NEDD8
Neural precursor cell-expressed developmentally downregulated protein 8 (NEDD8) plays a critical role in various cellular functions in cancers. Additionally, NEDD8-activating enzyme (NAE) is an essential component of the NEDD8 conjugation pathway. Besides, NEDD8-activating enzyme controls the activity of the cullin-RING subtype of ubiquitin ligases, thereby regulating the turnover of a subset of proteins upstream of the proteasome. NEDD8 is an ubiquitin-like modifier most well-studied for its role in activating the largest family of ubiquitin E3 ligases, the cullin-RING ligases. SENP8 is a cysteine protease. SENP8 selectively interacts with NEDD8 and not ubiquitin, and also plays a redundant role in proteolytic processing of the precursor form of NEDD8 in conjunction with ubiquitin C-terminal hydrolase isozyme 3 (UCHL3). Therefore, NEDD8 activating enzyme (NAE) plays a critical role in various cellular functions in cancers.
ZM223
In this study, Ma H, et al found that ZM223 exhibited antitumor activities with IC50 values in nanomolar range. ZM223 shows excellent anticancer activity against HCT116 colon cancer cells with an IC50 value of 100 nM. Moreover, ZM223 causes a dose-response decrease in the level of NEDD8 and an increase in the downstream UBC12 protein. Besides, ZM223 inhibits NEDD8 protein and subsequently elevates UBC12 level.