Non-Homologous End-Joining (NHEJ) is the important and crucial pathway for the repair of DNA double strand breaks (DSBs) in human cells. The NHEJ pathway is frequently upregulated in solid tumors as a compensatory mechanism for a separate DSB repair defect or for innate genomic instability. Moreover, NHEJ also reduces the efficacy of cancer treatment modalities that rely on the introduction of DSBs. Hence, inhibition of the NHEJ pathway can modulate a radiation- or chemo-refractory disease presentation.
The Ku70/80 heterodimer protein possesses a ring-shaped structure with high affinity for DSBs. Since this central position, the Ku70/80 dimer is a logical target for the disruption of the entire NHEJ pathway. Surprisingly, specific inhibitors of the Ku70/80 heterodimer are currently not available.
Researchers design a selected potential inhibitor STL127705, which has a capable of disrupting the binding of Ku70/80 to DNA substrates. Moreover, STL127705 also impairs Ku-dependent activation of another NHEJ factor, the DNA-PKCS kinase.
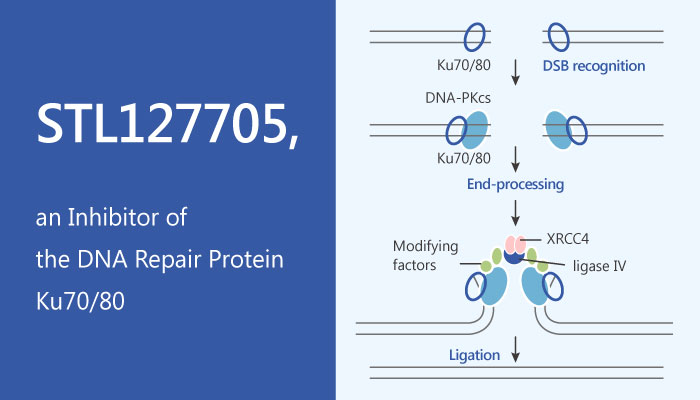
STL127705 (Compound L) is a Ku 70/80 heterodimer protein inhibitor, inhibits Ku70/80-DNA interaction, with an IC50 of 3.5 μM. STL127705 also inhibits Ku-dependent activation of DNA-PKCS kinase (IC50, 2.5 μM). Furthermore, researchers’ exposes cultures of the human glioblastoma cell line SF-767 glioblastoma cells to increasing concentrations of STL127705. With the increasing doses of STL127705, the autophospho-rylation of DNA-PKCS lowers more. STL127705 is able to inhibit the Ku-dependent activation of the DNA-PKCS kinase in human cells.
Hopefully, STL127705 synergistically sensitized human cell lines to radiation treatment, indicating a clear potential to diminish DSB repair. STL127705 is able to be utilized as a lead-generating platform for the development of a novel anti-cancer agent.