The spindle assembly checkpoint (SAC) is important for cell survival. Its inactivation generates lethal genomic instability in cancer cells. Inhibition of SAC signaling through targeting of Mps1 has provided an indication of the feasibility of such an approach. Mps1 is also an essential, dual-specificity kinase that functions as an important guardian of the fidelity of chromosome segregation. It is critical for the recruitment of SAC proteins to unattached kinetochores, mitotic checkpoint complex formation, and thus APC/C inhibition. Mps1 is also required for chromosome alignment and error correction. Inhibition of Mps1 activity causes cells to prematurely exit mitosis with unattached chromosomes. This results in severe chromosome missegregation, aneuploidy, and ultimately cell death.
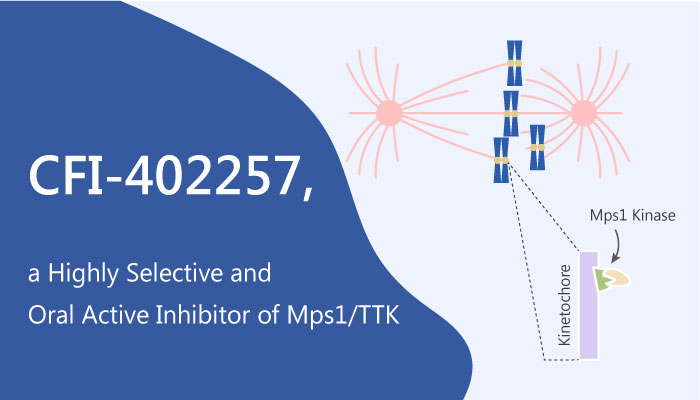
Liu et al identified CFI-402257, a potent, selective, and oral inhibitor of Mps1. It inhibited Mps1 with an IC50 of 1.2 nM and a Ki of 0.09 nM. This inhibitor also shows strong antineoplastic activity on a broad panel of human cancer-derived cell lines. Additionally, it causes effects consistent with depletion or inhibition of Mps1. CFI-402257 treatment leads to SAC inactivation, chromosome missegregation, aneuploidy, and ultimately cell death. CFI-402257 has demonstrated efficacy as a monotherapy in cancer cell line and patient-derived xenograft (PDX) models across various indications. It also demonstrated antitumor activity in a platinum-resistant PDX model of high-grade serous ovarian cancer. CFI-402257 induces genomic instability and apoptotic cell death and therefore could promote tumor immunity. CFI-402257 is a potential small-molecule inhibitor for the treatment of cancer with a mechanism of action that is consistent with the potent and selective inhibition of Mps1.