Chronic myeloid leukemia is a myeloproliferative disorder characterized by the presence of the oncogenic fusion protein BCR-ABL1. ABL1, one of the fusion partners in the BCR-ABL1 protein, contains an allosteric myristoylation binding site. Especially, the proteolysis targeting chimera (PROTAC) technology provides a unique toolkit for probing the non-kinase roles of proteins in a cellular context without resorting to genetic modification of the system. Particularly, PROTACs are heterobifunctional molecules that recruit an E3 ligase to a target protein. In this study, George M. Burslem, et al further investigate the potential of a new series of PROTACs, utilizing an allosteric inhibitor (GNF-5) to degrade BCR-ABL1 protein.
ABL1 tyrosine kinase inhibitors (TKIs) effectively reduce kinase activity and downstream signaling of BCR-ABL1. George M. Burslem, et al developed a series of PROTACs. In particular, PROTACs allosterically target BCR-ABL1 protein and recruit the E3 ligase Von Hippel Lindau (VHL). Consequently, this effect results in ubiquitination and subsequent degradation of the oncogenic fusion protein. Typically, the lead compound is GMB-475. Moreover, in both human CML K562 cells and murine Ba/F3 cells expressing BCR-ABL1, GMB-475 induces rapid proteasomal degradation and inhibition of downstream biomarkers. Notably, GMB-475 inhibits the proliferation of certain clinically relevant BCR-ABL1 kinase domain point mutants. Importantly, GMB-475 reduces viability and increases apoptosis in primary chronic myeloid leukemia CD34+ cells, with no effect on healthy CD34+ cells. Furthermore, GMB-475 degrades BCR-ABL1 and reduces cell viability in primary chronic myeloid leukemia stem cells.
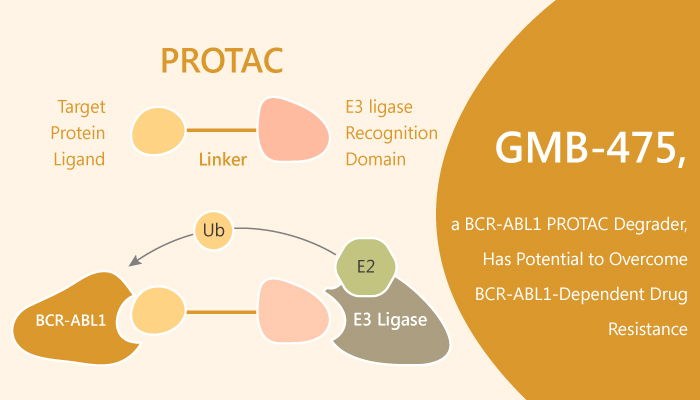
Together, these findings suggest that targeting BCR-ABL1 in chronic myeloid leukemia by PROTAC-mediated targeted protein degradation. In addition, GMB-475 reduces cell viability, induces apoptosis, and degrades BCR-ABL1 in primary chronic myeloid leukemia patient stem/progenitor cells.