YEATS domain is a histone acylation reader in health and diseases. There are four YD‐containing genes in humans (MLLT1, YEATS2, MLLT3, and YEATS4). YEATS domain (YD) containing proteins are an emerging class of epigenetic targets in drug discovery. Of these, YEATS2 is a YEATS domain-containing protein conserves from fly to human. Besides, YEATS2 is a stoichiometric subunit of the ADA Two A-Containing (ATAC) histone acetyltransferase complex in transcription regulation, stress signaling and mitotic progression. Alternatively, YEATS4 is a stoichiometric component of the SRCAP (SNF2-related CREBBP activator protein) and Tip60/p400 chromatin remodeling complexes.
MLLT1 (ENL/LTG19) is one of a number of fusion gene partners with the MLL oncogene in 11q23 translocations in human leukemia and encodes a transcriptional regulator of unknown function. Leukemias bearing MLL translocations may be myeloid or lymphoid or bear mixed-lineage properties; however, those bearing MLLT1/3 translocations are predominantly lymphoid, suggesting that MLLT may influence the leukemic phenotype. In particular, MLLT1/3 in essential developmental processes and suggest that expression patterns of MLL fusion partners may influence the lineage of MLL-associated leukemias.
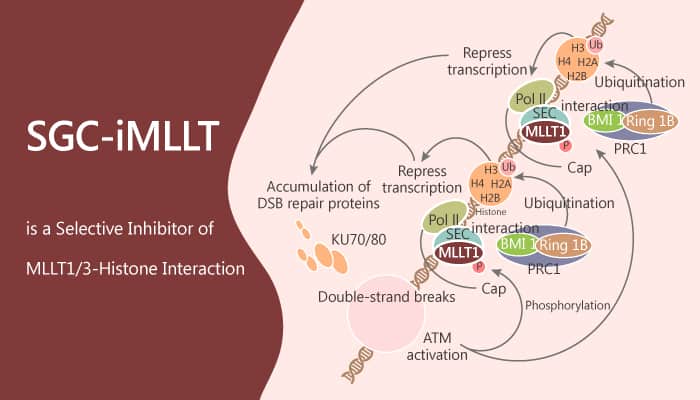
In this study, Dr. Moses Moustakim, et al report the discovery and characterization of the first small‐molecule chemical probe, SGC-iMLLT. Especially, SGC-iMLLT is a potent and selective inhibitor of MLLT1/3-histone interactions with an IC50 of 0.26±0.09 uM. Moreover, SGC-iMLLT has excellent selectivity over other human YD proteins (YEATS2/4) and bromodomains. Furthermore, SGC-iMLLT will enable researchers to design the first biological experiments exploring MLLT1/3 YD inhibition. Researchers also investigate the metabolic stability of SGC-iMLLT in primary human hepatocytes. As a result, SGC-iMLLT shows moderate metabolic resistance (t1/2 53 min, 48 % remaining after 60 min), with a primary process for metabolism being N demethylation.