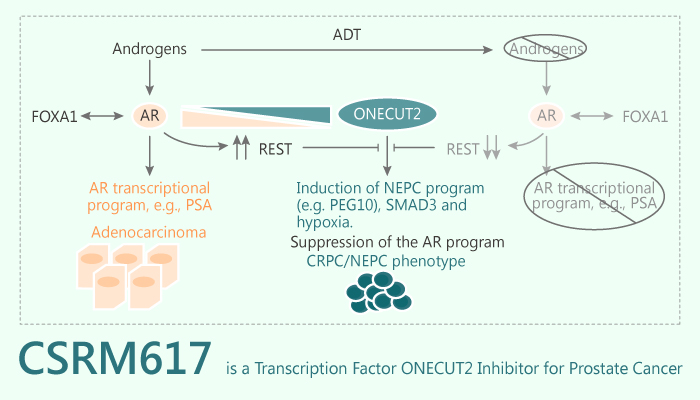
The transcription factor ONECUT2 (OC2) is a master regulator of androgen receptor networks in mCRPC (metastatic castration-resistant prostate cancer ). OC2 acts as a survival factor in mCRPC models.
OC2 is a potential drug target in the metastatic phase of aggressive PC.
CSRM617, a well-tolerated novel small-molecule inhibitor of the OC2, binds to the OC2-HOX domain directly with a Kd of 7.43 uM in SPR assays
In vitro, CSRM617(20 nM-20 μM; 48 hours) inhibits a panel of PC cell lines growth, including 22Rv1 cells. Meanwhile, CSRM617(20 μM; 72 hours) induces 22Rv1 cell apoptosis via increasing cleaved Caspase-3 and PARP expression. However, CSRM617 has slight effects on OC2 depleted with shRNA or siRNA 22Rv1 cells, thus, cells express high levels of OC2 are more responsive to CRSM.
Then, the authors choose a directly-regulated OC2 gene, PEG10. CSRM617 (4-16 hours) decreases PEG 10 mRNA expression in a time-dependent manner. Therefore, CSRM617 is a specificity regulator for OC2.
In vivo, Firstly, the authors subcutaneously implant 22Rv1 cells to nude mice. CSRM617 reduces tumor volume and weight significantly. At the same time; CSRM does not affect mouse weight, it well tolerated in the mouse.
Secondly, the authors intracardially inject luciferase-tagged 22Rv1 cells in SCID mice. After two days, the mouse is treated daily (50 mg/Kg) with CSRM617 or vehicle. CSRM617 elicits a significant reduction in the onset and growth of diffuse metastases. As a result, CSRM617 is effective against PC metastases.
Thirdly, they measure PEG10 protein levels in tumors in both CSRM617 and control groups. CSRM617 significantly down-regulates PEG10 expression in tumors from experimental group mice. PEG 10 not only is an effective metric for bioactivity of CSRM617 in vitro but also in Vivo.
Lastly, the authors intracardially inject luciferase-tagged 22Rv1 cells in SCID mice to develop metastases. CSRM617 makes a significant reduction of diffuse metastases, quantitatively down-regulating PEG10 protein levels in tumors in both the experimental and control groups. From these results, CSRM617 has the treating efficacy in tumor metastasis mouse.
In summary, CSRM617 is effective against prostate cancer (PC) in vitro and Vivo.
Reference:
Rotinen M, et al. Nat Med. 2018 Dec;24(12):1887-1898.