Mesenchymal stem cells (MSCs) are multipotent progenitor cells that play a critical role in tissue regeneration. MSCs play an important role in matrix remodeling, fibroblast activation, angiogenesis, and immunomodulation. MSCs are an integral part of fibrovascular networks that form in developing tissues and tumors. A multitude of soluble proteins regulates the engraftment and function of MSCs in tissue niches.
PDGF is a crucial regulator of MSC growth, proliferation, survival, and chemotaxis.
Platelet-derived growth factor-BB (PDGF) is essential for MSC recruitment to nascent vessels and maturation into perivascular cells. PDGF interacts with PDGFRα and β tyrosine kinase receptors that dimerize for activation of intracellular signaling. After development, MSCs primarily express PDGFR-β, which, with its ligand PDGF-B, plays a critical role in mediating the tropism and differentiation during vascular remodeling.
In order to evaluate whether this pathway was important for the effect of gastric cancer-derived mesenchymal stem cell-conditioned medium (GC-MSC-CM) on gastric cancer cells, Feng Huang, et al pretreated SGC-7901 cells with PDGFRβ inhibitor SU16f before co-culture with GC-MSCs. The results show that SU16f significantly abolishes PDGFRβ activation in SGC-7901 by GC-MSC-CM. Moreover, Western blot analysis reveals that SU16f pretreatment results in the upregulation of E-cadherin and downregulation of N-cadherin, Vimentin, and α-SMA. SU16f significantly decreases the enhanced migratory ability of SGC-7901 cells by GC-MSC. In addition, SU16f pretreatment leads to downregulation of p-AKT, Bcl-xl, and Bcl-2 levels and upregulation of Bax expression in SGC-7901 cells by GC-MSC-CM. Cell colony formation assay and MTT assay show that SU16f pretreatment inhibits the promoting role of GC-MSC-CM in SGC-7901 cell proliferation.
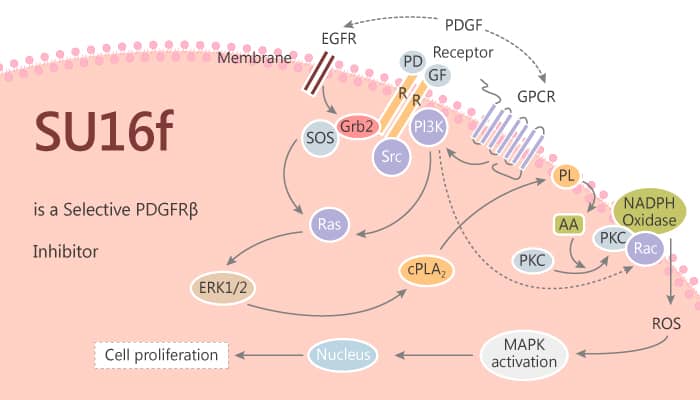
All in all, PDGFRβ signaling is critical for GC-MSC regulation of migration and proliferation in gastric cancer cells. Neutralization of PDGFR-β by SU16f blocks the promoting role of GC-MSC conditioned medium in gastric cancer cell proliferation and migration.