Post-translational modification of histones by histone-modifying enzymes represents a key element of epigenetic regulation. Epigenetic reader domains are structurally diverse. They include bromodomains, PHD domains, methyl-lysine- and/or methyl-arginine-binding domains, frequently co-locating within the same proteins. By interfering with the function of specific domains, selective chemical probes represent powerful tools to dissect the biological functions of these multi-valent epigenetic regulators. Nuclear receptor-binding SET domain 3 (NSD3) is a multi-domain protein that exists in three isoforms. Both NSD3 long and Whistle isoforms contain the SET domain, as well as several chromatin reader domains, including PHD and PWWP domains. The NSD3-short isoform contains only the first PWWP domain and a recent study indicated that it serves as an adapter protein, this function is dependent on the PWWP1 domain and required for the viability of acute myeloid leukemia (AML) cells. In this study, BI-9321 is a chemical probe for the PWWP1 domain of NSD3.
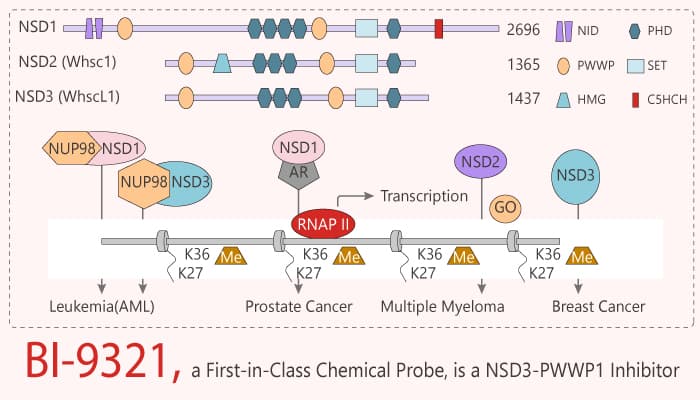
BI-9321 is a first-in-class chemical probe. it targets the methyl-lysine binding site of the PWWP1 domain with sub-micromolar in vitro activity and cellular target engagement at 1 µM. Meanwhile, it is a potent, selective NSD3-PWWP1 domain antagonist, with a Kd value of 166 nM. BI-9321 is inactive against NSD2-PWWP1 and NSD3-PWWP2. BI-9321 specifically disrupts histone interactions of the NSD3-PWWP1 domain with an IC50 of 1.2 μM in U2OS cells. In addition, BI-9321 potentiates the effects of JQ1 in the MOLM-13 cell line. Furthermore, it downregulates Myc messenger RNA expression and reduces proliferation in MOLM-13 cells.
In summary, BI-9321 will enable the scientific community to determine effects on cells on NSD3-PWWP1 inhibition. NSD3-PWWP1 domain antagonism in combination with other targeted therapies might be relevant to cancer therapy.
Reference:
Böttcher J, et al.2019 Aug;15(8):822-829.