Stimulator of interferons genes (STING) is an adaptor protein. It plays a critical role in the secretion of type I interferons and pro-inflammatory cytokines in response to cytosolic nucleic acid. STING is also an endoplasmic reticulum-associated homodimeric protein. It is the receptor for cGAMP. The cGAMP is a second messenger from the cGAMP synthase. cGAMP binding to STING induces a pronounced conformational change. In addition, activation of STING by cGAMP triggers downstream signaling events initially via interactions of the closed-form complex with TBK1 kinase, and later by the transcription factors IRF3 and NFkB, culminating in increased synthesis and secretion of type I interferons and proinflammatory cytokines. In this study, MSA-2 is a non-nucleotide STING agonist that preferentially targets tumor tissue. Moreover, it shows antitumor activity and stimulates interferon-β secretion in tumors, induces tumor regression with durable antitumor immunity.
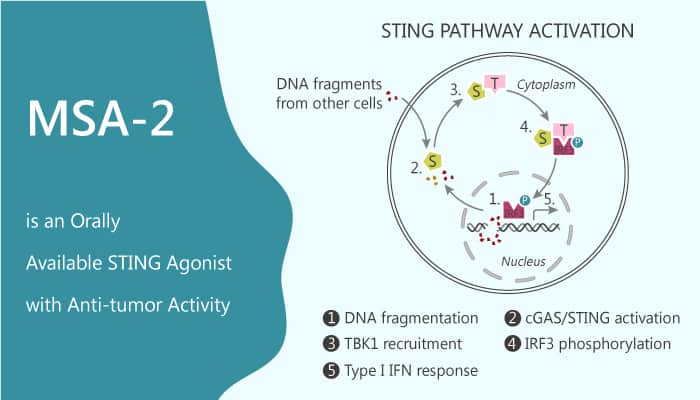
MSA-2 shows EC50s of 8.3 and 24 μM for human STING isoforms WT and HAQ, respectively. Furthermore, MSA-2 (IT, SC, or PO routes) also exhibits dose-dependent antitumor activity and induces complete tumor regressions in 80 to 100%. MSA-2 effectively inhibits tumor growth induced substantial elevations of IFN-β, interleukin-6 (IL-6), and TNF-α in the tumor. In addition, MSA-2 enhances in vivo antitumor activity of the anti-PD-1 antibody.
In summary, MSA-2 is an orally bioavailable STING agonist with in vivo antitumor activity. It shows antitumor activity and stimulates interferon-β secretion in tumors. MSA-2 also induces tumor regression with durable antitumor immunity and synergizes with anti-PD-1 in syngeneic mouse tumor models. MSA-2 is a valuable tool in the endeavor to discover and design human STING agonists, which are suitable for systemic administration in the clinic.
Reference:
Pan BS, et al. Science. 2020;369(6506):eaba6098.