Epidermal growth factor (EGF) receptor (EGFR), is the prototype of the EGFR family that also includes ErbB2/HER2/Neu, ErbB3/HER3, and ErbB4/HER4. EGFR is commonly upregulated in cancers such as in non-small-cell lung cancer, metastatic colorectal cancer, glioblastoma, head and neck cancer, pancreatic cancer, and breast cancer. Various mechanisms mediate the upregulation of EGFR activity, including common mutations and truncations to its extracellular domain. These EGFR aberrations over-activate downstream pro-oncogenic signaling pathways, including the RAS-RAF-MEK-ERK MAPK and AKT-PI3K-mTOR pathways. In this study, is a potent and selective EGFR tyrosine kinase inhibitor (IC50=0.7 μM). AG 494 inhibits EGF- or serum-induced cell proliferation. It inhibits the autophosphorylation of EGFR, ErbB2, HER1-2, and PDGF-R with IC50s 1.1, 39, 45, and 6 μM, respectively. AG-494 blocks Cdk2 activation and inhibits EGF-dependent DNA synthesis.
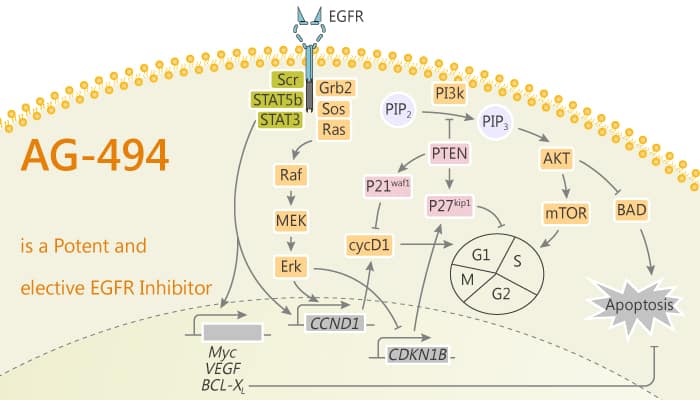
AG494 is specific in inhibiting HERl kinase activity. It is also quite effective in inhibiting platelet-derived growth factor receptor autophosphorylation. AG494 inhibits the phosphorylation of poly-Glu4-Tyrl catalyzed by HER1 20-fold more potently than HER1-2. AG 494 retains its full inhibitory activity on EGF‐dependent DNA synthesis measured by [3H]thymidine uptake, even when added 20 h subsequent to EGF addition. Since AG 494 inhibits not only EGF‐ but also serum‐dependent cell growth and LPA‐ or TPA‐dependent DNA synthesis. AG 494 inhibits in a dose‐dependent manner of Cdk2 EGF‐dependent activation with an IC50 ∼15 μM. In addition, AG-494 significantly prevents NF-kB activation in silica-stimulated cells and also reduces NF-kB activation in H2O2-treated cells.
In summary, AG-494 is a potent and selective EGFR tyrosine kinase inhibitor. It inhibits the autophosphorylation of EGFR, ErbB2, HER1-2, and PDGF-R. AG-494 also blocks Cdk2 activation and inhibits EGF-dependent DNA synthesis.
References:
Osherov N, et al. FEBS Lett. 1997;410(2-3):187-190.; Osherov N, et al.. J Biol Chem. 1993;268(15):11134-11142.