NUT midline carcinoma (NMC) is a rare, aggressive subtype of squamous carcinoma. This kind of cancer is usually driven by BRD4-NUT fusion oncoprotein. BRD4, a BET protein, binds to chromatin through its two bromodomains. At the same time, NUT recruits the p300 HAT to activate oncogenic target genes transcription. BET selective bromodomain inhibitors demonstrate on-target activity in NMC patients, but with limited efficacy. P300, like BRD4, contains a bromodomain. Scientists have shown that combining selective p300/CBP and BET bromodomain inhibitors, GNE-781 and OTX015, respectively, induces cooperative depletion of MYC and synergistic inhibition of NMC growth. Especially, in this, blog, we would like to introduce a novel dual p300/CBP and BET bromodomain selective inhibitor NEO2734.
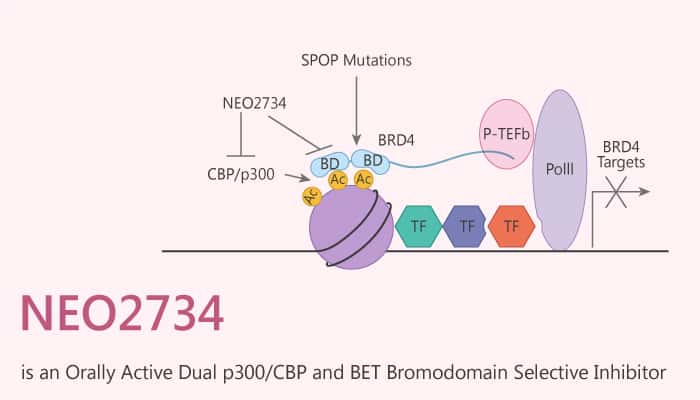
NEO2734 (EP31670) is an orally active and dual p300/CBP and BET bromodomain selective inhibitor, with IC50 values of <30 nM for both p300/CBP and BET bromodomains.
In NUT carcinoma patient cell lines (TC-797 and PER-403), NEO2734 (1 μM) treatment results in greater loss of MYC protein. Moreover, NEO2734 (1 μM) induces differentiation and G1-phase cell cycle arrest. NEO2734 (1 μM) also rapidly induces squamous differentiation in NMC cell lines, and expression of the terminal squamous differentiation marker, involucrin, or keratins.
In Mice (PER-403 and 14169 models), NEO2734 (5, 8, 10 mg/kg, orally) once daily for 28 days inhibits growth and prolongs survival in pre-clinical xenograft models. In the treatment group, two of the three mice treated with NEO2734 were alive by day 100. The compound provided markedly improved survival compared with EP, i-BET-762, and even iBET-762+EP by day 100 following initiation of treatment.
Not only that, NEO2734 is active in both hotspot mutant (F133V) and non-hotspot mutant (Q165P) PCa cells in vitro and in vivo.
In total, NEO2734 is a potential lead compound for NUT midline carcinoma therapy.
Reference: