Targeting protein degradation refers to small molecule induces ubiquitination and degradation of disease targets. These small molecules simultaneously recruit both a ubiquitin E3 ligase and the target protein, then to be ubiquitinated. Therefore, PROTACs represent a functional application of chemically induced protein dimerization. BRD4 acts as an attractive target for anticancer therapy. However, BRD4 inhibitors treatment leads to BRD4 protein accumulation. The reversible nature of inhibitors binding to BRD4 may limit the efficacy of BRD4 inhibitors. To address these problems, a protein degradation strategy based on the PROTAC technology has been developed to target BRD4.
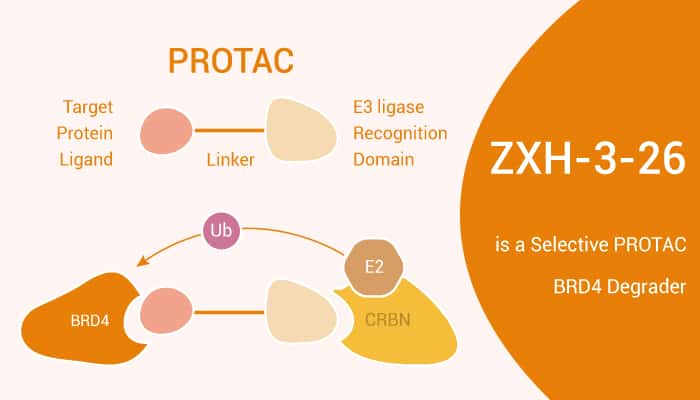
In this study, researchers present the synthesis and biological evaluation of ZXH-3-26. ZXH-3-26 is a selective PROTAC BRD4 degrader. Gratifyingly, ZXH-3-26 shows excellent inhibitory activity against BRD4, and high anti-proliferative potency. Cellular degradation assays show that ZXH-3-26 exhibits a DC50/5h ~ 5 nM comparable to the best pan-BRD degrader dBET6. ZXH-3-26 shows activity exclusively on the first bromodomain of BRD4, and spares degradation of BRD2 or 3 at concentrations 10 μM, while dBET6 and MZ1 controls show activity on most bromodomains. Immunoblot analysis confirms that ZXH-3-26 degrades endogenous BRD4 with comparable efficacy compared to the best pan-BET degrader dBET6, while being inactive on BRD2 and BRD3.
ZXH-3-26 engages CRBN in a conformation distinct. Moreover, ZXH-3-26 thus demonstrates that binding to a distinct conformation can yield a highly selective degrader molecule and that selectivity can be achieved across highly homologous domains such as the bromodomains of BET proteins. Furthermore, ZXH-3-26 represents the first small molecule to allow pharmacologic targeting of BRD4 without significant inhibition or degradation of BRD2/3.
All of these results suggest that ZXH-3-26 is an efficacious BRD4 degrader for further investigation.