The bromodomain and extra-terminal (BET) family proteins consist of BRD2, BRD3, BRD4, and testis-specific BRDT members. They are epigenetic “readers” and play a key role in the regulation of gene transcription. BET proteins are attractive therapeutic targets for cancer and other human diseases. In this study, BETd-260 is capable of inducing degradation of BRD2, BRD3, and BRD4 proteins at 30–100 pM in the RS4;11 leukemia cells. Moreover, it also achieves an IC50 value of 51 pM in inhibition of the RS4;11 cell growth. In addition, BETd-260 induces rapid tumor regression of the RS4;11 xenograft tumors with no signs of toxicity in mice. In addition, BETd-260 potently suppresses cell viability and robustly induces apoptosis in HCC cells. BETd-260 reciprocally modulates the expression of several apoptotic genes in HCC cells, i.e., suppressing the expression of anti-apoptotic Mcl-1, Bcl-2, c-Myc, and XIAP, whereas increasing the expression of pro-apoptotic Bad.
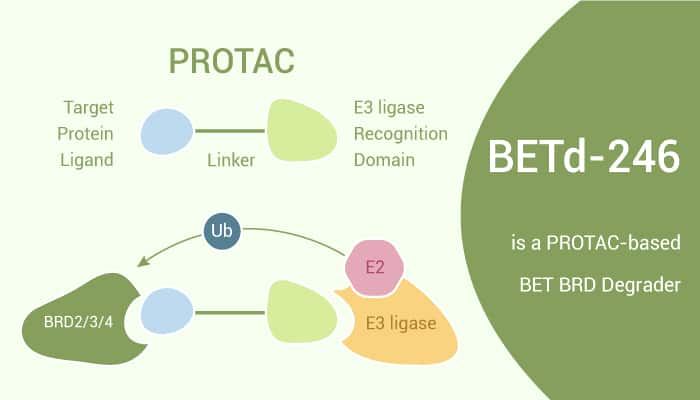
BETd-260 shows inhibitory activity against the growth of RS4;11 leukemia cells and MOLM-13 cells with IC50s of 51 pM and 2.2 nM, respectively. It also induces apoptosis in both RS4;11 and MOLM-13 cell lines at 3-10 nM. Moreover, it causes rapid tumor regression with a maximum of >90% regression in mice bearing RS4;11 xenograft tumors. Furthermore, it has no body-weight loss or other signs of toxicity in mice. In addition, BETd-260 degrades the BRD2, BRD3, and BRD4 proteins for more than 24 h, with robust cleavage of PARP and caspase-3. It also strong down-regulation of c-Myc protein in RS4;11 xenograft mice model.
In summary, BETd-260 is a highly potent, efficacious, and promising BET degrader. It warrants further evaluation as a potential new therapy for the treatment of human acute leukemia and other types of human cancer.
Reference:
Zhou B, et al. J Med Chem. 2018 Jan 25;61(2):462-481.; Zhang H, et al. Front Oncol. 2020;9:1471. Published 2020 Jan 14.