DNA2 nuclease possesses 5′ flap endonuclease and 3′-5′ helicase activities. It has an important role in DNA damage repair, homologous recombination, and DNA replication. The nuclease activity of DNA2 has several well-documented cellular functions. However, the biological role of its helicase activity remains enigmatic. DNA2 mediates the resection of the 5′ strand at DNA DSB ends, an early step in HR, and also at stalled and regressed replication forks. In this study, DNA2 is overexpressed in pancreatic cancers. It is one of the deadliest and more aggressive forms of human cancers, where mutations in the KRAS are present in 90–95% of cases. In addition, depletion of DNA2 significantly reduces pancreatic cancer cell survival and xenograft tumor growth. Thus, DNA2 inhibition is a potential therapeutic strategy. In this study, NSC-105808 is a potent, specific DNA2 nuclease inhibitor, with IC50s of 2.0 and 1.49 μM for yeast and human DNA2.
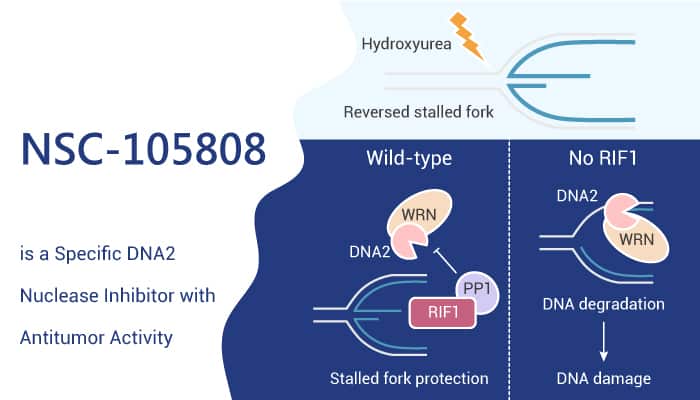
NSC-105808 specifically inhibits the nuclease activity of DNA2. It also inhibits HR repair, double-strand DNA breaks (DSB) end resection, and suppresses the proliferation of cancer cells. Moreover, NSC-105808 interferes with DNA end resection and HR in cells and suppresses the cisplatin sensitivity of FANCD2−/− cells. It significantly attenuates the accumulation of Camptothecin-induced p-RPA, suggesting an impairment of DSB end resection. Furthermore, NSC-105808 reduced p-RPA accumulation in the chromatin fraction with a concomitant enhancement in the γ-H2AX level, reflective of an accumulation of unrepaired DSBs.
In summary, the inhibition of DNA2 nuclease is a therapeutic strategy targeting replication stress in cancer cells. NSC-105808 is a potent, specific DNA2 nuclease inhibitor.
Reference:
Kumar S, et al. Oncogenesis. 2017;6(4):e319. Published 2017 Apr 17.