Tamoxifen is a nonsteroidal agent and competitively binds to estrogen receptors (ER). This compound itself is a prodrug and it has a relatively little affinity for ER. However, tamoxifen can transform into active metabolites 4-hydroxytamoxifen and N-desmethyl-4-hydroxytamoxifen (endoxifen). Besides, these metabolites exhibit 30–100 times more affinity with the estrogen receptor than tamoxifen itself.
FLTX1 is a fluorescent Tamoxifen derivative.
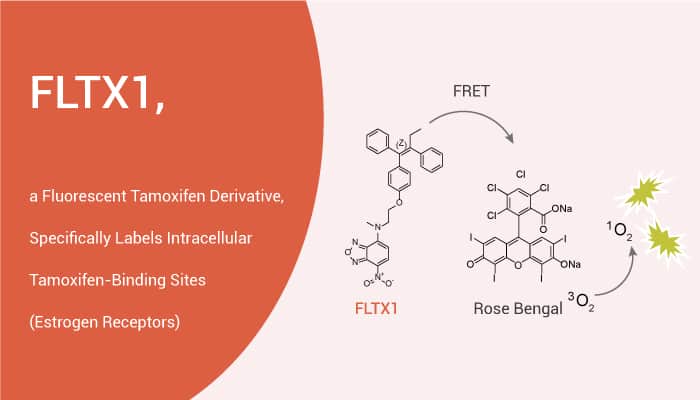
It can specifically label intracellular Tamoxifen-binding sites (estrogen receptors) under permeabilized and non-permeabilized conditions. FLTX1 exhibits the potent antiestrogenic properties of Tamoxifen in breast cancer cells. But FLTX1 is devoid of the estrogenic agonistic effect on the uterus.
In MCF7 cells, FLTX1 reduces MCF7 cell proliferation in a dose-dependent manner. Besides, it being significantly more effective than Tamoxifen (Tx) already at 0.1 μM. Pretreatment with FLTX1 counteracts the increase in cell growth induced by E2 down to the vehicle level. Additionally, FLTX1 competitively displaces the [3H] E2 binding to rat uterine estrogen receptors (ER) rat uterus cytosol, with an IC50 of 87.5 nM.
Agonistic and antagonistic ER-mediated transcriptional activity is assayed using transiently transfected MCF7 cells and stably transfected T47D-KBluc cell line. Tx significantly increases transcriptional activity in both cell lines in a dose-dependent manner. The concentration of FLTX1 exists in a dose-dependent manner from 10 nM -1 μM in MCF7 and 10 nM- 1 μM in T47D-KBluc cells, respectively. However, FLTX1 is devoid of the ability to stimulate luciferase transcriptional activity in MCF7 cells.
FLTX1 has antiestrogenic activity. As a result, FLTX1 decreases E2-induced luciferase activity in a dose-dependent manner. The IC50 values are 1.74 μM and 0.61 μM in MCF7 and T47D-BKluc cells, respectively.
In immature female CD-1 mice and Sprague–Dawley rats, FLTX1 fails to alter basal proliferating cell nuclear antigen immunoreactivity.
In conclusion, FLTX1 can specifically label intracellular Tamoxifen-binding sites and exhibits potent antiestrogenic properties.
[1]. Marrero-Alonso J, et, al. Eur J Pharm Biopharm. 2013 Nov;85(3 Pt B):898-910.
[2]. Morales A, et, al. Methods Mol Biol. 2016;1366:163-173.