Autophagy-targeting chimera (AUTAC) is a technology that selectively degrades intracellular proteins and intracellular debris through the autophagy pathway. An AUTAC molecule contains a degradation tag (guanine derivative) and a ligand of the protein of interest (POI) to provide target specificity. AUTAC can selectively recognize and degrades a broad range of cellular proteins including their misfolded aggregates. What’s more, the AUTAC system has a similar design to the PROTAC technology. Both AUTAC and PROTAC molecules function via ubiquitination. However, instead of tethering the POIs to an E3 ligase subunit and triggering K48 polyubiquitination, AUTAC molecules trigger K63 polyubiquitination, leading to degradation of the target POI. Besides, AUTAC molecules are capable of degrading cellular organelles such as damaged mitochondria.
 Currently, AUTAC technology has been successfully applied to degrade many proteins such as MetAP2, FKBP12, BET, and TSPO. A recent study has reported that AUTAC molecules can promote the phagocytosis of broken or impaired mitochondria, which are related to diseases of aging. It is foreseeable that AUTAC molecule specifically degrades intracellular proteins or disrupts organelles through selective autophagy, which has the potential to become one of the most promising strategies for the treatment of diseases related to organ damage.
Currently, AUTAC technology has been successfully applied to degrade many proteins such as MetAP2, FKBP12, BET, and TSPO. A recent study has reported that AUTAC molecules can promote the phagocytosis of broken or impaired mitochondria, which are related to diseases of aging. It is foreseeable that AUTAC molecule specifically degrades intracellular proteins or disrupts organelles through selective autophagy, which has the potential to become one of the most promising strategies for the treatment of diseases related to organ damage.
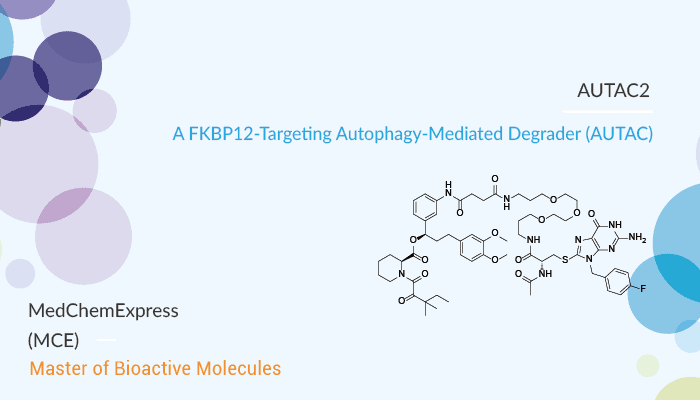
AUTAC2 is an AUTAC. AUTAC2 can degrade FK506-binding protein (FKBP12). FKBP12 binds the immunosuppressant molecule Tacrolimus (originally designated FK506) in vivo, which is for autoimmune disorders. Importantly, AUTAC2 contains an FBnG (p-Fluorobenzyl Guanine) and an SLF (c ligand of FKBP) moiety. The warhead synthetic ligand of FKBP (SLF) is a non-covalent targeting ligand of FKBP12. At an AUTAC2 concentration of 10 μM, resulting in significant silencing of FKBP12 in HeLa cells, demonstrating the potential of AUTACs for protein silencing.
All in all, AUTAC2 is an FKBP12-targeting autophagy-mediated degrader (AUTAC).
References:
Ji CH, et, al. Nat Commun. 2022 Feb 16;13(1):904.