In this article, we will introduce a long-acting recombinant human IL-7, Efineptakin alfa. Efineptakin alfa is a potent T-cell amplifier, with a homodimeric IL-7 fused to the hybridizing IgD/IgG4 immunoglobulin domain.
Efineptakin alfa supports the proliferation and survival of CD4+ and CD8+ cells in both humans and mice. Patients with glioblastoma (GBM) are treated with radiotherapy (RT) or temozolomide (TMZ). However, these treatments may prolong systemic lymphopenia. Efineptakin alfa (10 mg/kg) mitigates RT-related lymphopenia, increases cytotoxic CD8 T lymphocytes systemically and in the tumor, and improves survival in orthotopic glioma-bearing mice.
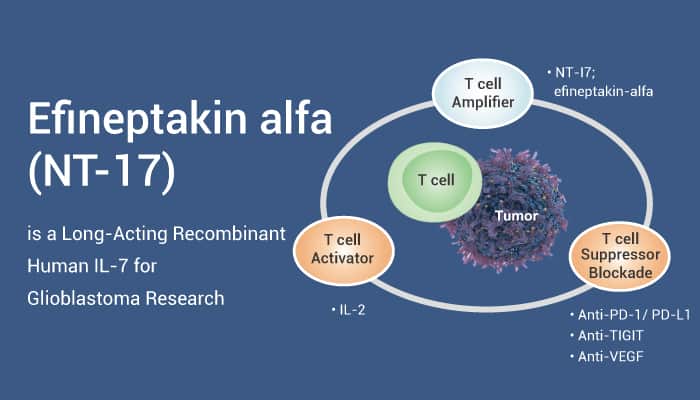
In the C57BL/6 mice bearing intracranial tumors (GL261 or CT2A) model, Efineptakin alfa (10 mg/kg, IM, single) combined with SLC-3010 (1.8 mg/kg, IV, single) inhibits tumor growth in MC38-bearing mice. SLC-3010 is a T cell activator (hIL-2/TCB2c complex). SLC-3010 can selectively activate T cells while disfavoring Treg activation. The combination of NT-I7 with SLC-3010 enhanced the antitumor response. It leads to an increased number and frequency of CD8+ T cells as well as granzyme B expression in the tumor. It Increased T lymphocytes in the lymph nodes, thymus, and spleen, enhancing IFNγ production. Additionally, it decreases Tregs in the tumor which was associated with a significant increase in survival. The treatment also enhances central memory and effector memory of CD8 T cells in lymphoid organs and tumors. At the same time, it decreases progenitor cells in the bone marrow.
Additionally, NT-I7 increased the frequency of PD-1+TCF-1+TOX– stem-like CD8+ T cells in the draining lymph node. Meanwhile, SLC-3010 significantly increased the number of PD-1+ CD8+ T cells in the tumor. All the data suggests that NT-I7 combined with other immunotherapies such as IL-2 to enhance the anti-tumor response.
Reference
[1]. Campian JL, et al. Clin Cancer Res. 2022 Mar 15;28(6):1229-1239.
[2]. Seungtae Baek, et al. 2022 AACR Abstract #4199.