In this article, we introduce a specific inhibitor of transforming growth factor-β (TGF-β) — PBrP. It suppresses TGF-β-induced cellular responsiveness by displacing type II TGF-β receptor (TβRII) from the plasma membrane into the lysosomal degradation pathway.
TGF-β is a member of a large family of evolutionarily conserved pleiotropic cytokines. It also includes bone morphogenetic proteins, activins, and nodal. Dysregulation of TGF-β signaling plays an important role in many diseases such as tissue fibrosis and cancer. Firstly, TGF-β exerts protective or tumor-suppressive effects on normal epithelial cells and during the early growth-sensitive stages of tumorigenesis. Secondly, the binding of the TGF-β to TβRII triggers the canonical pathway. Then activated TβRI phosphorylates receptor-activated Smads, which subsequently form a heterocomplex with Smad4. Finally, the Smad complex is then rapidly translocated to the nucleus to regulate target gene activity.
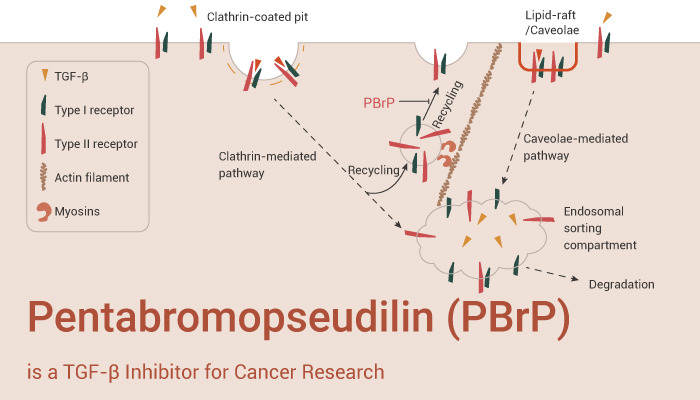
Pentabromopseudilin (PBrP) is a TGF-β inhibitor and can be used for the research of cancer.
Firstly, Pentabromopseudilin (PBrP) is a marine antibiotic. It is isolated from the marine bacteria Pseudomonas bromoutilis and Alteromonas luteoviolaceus. Secondly, PBrP exhibits antimicrobial, anti-tumor, and phytotoxic activities. Thirdly, PBrP is a reversible and allosteric inhibitor of myosin Va (MyoVa). Furthermore, PBrP also is a potent inhibitor of transforming growth factor-β (TGF-β) activity.
In vitro, Pentabromopseudilin (PBrP) (0.01-1 μM, 6 h) prevents TGF-β-induced Smad protein phosphorylation and nuclear translocation. First, PBrP (0.5 μM, 6 h) inhibits TGF-β-stimulated transcriptional responses. Second, PBrP (0-1 μM, 6 h) inhibits TGF-β-induced EMT in A549 cells. Then, PBrP (0.2 μM, 20 h) suppresses TGF-β-induced cell migration. Finally, PBrP (0.5 μM, 0, 1, 3 h) blocks TGF-β signaling by enhancing the degradation of TβRII via caveolae.
Reference
[1] Shih-Wei W, et al. J Enzyme Inhib Med Chem. 2018;33(1):920-935.
[2] Chung CL, et al. Int J Mol Sci. 2019;20(16):3913.