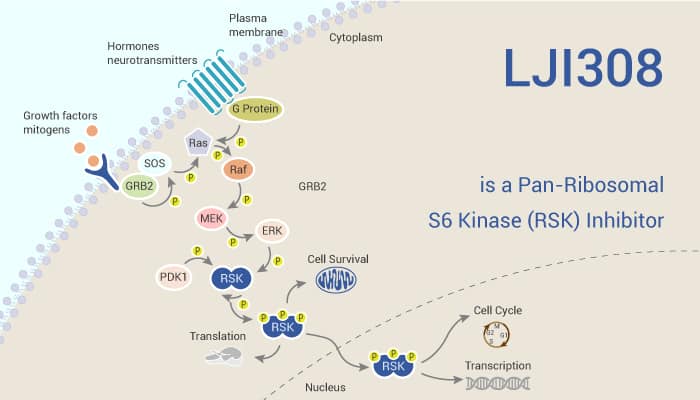
The cold shock protein Y-box binding protein-1 (YB-1) is a multifunctional protein. It shows relations in cell proliferation, survival, and stress response. Through translation and transcription of key proteins and regulation of DNA repair, YB-1 performs its functions in different cancers. YB-1 displays overexpression in many tumor entities, especially glioblastoma and breast, cervical, colorectal, and prostate carcinoma in association with chemotherapy or radiation therapy resistance. Ionizing radiation, receptor tyrosine kinase ligands, and mutation in KRAS gene stimulate the activation of YB-1. YB-1 accelerates the repair of IR-induced DNA double-strand breaks (DSBs). Ribosomal S6 kinase (RSK) is the main kinase inducing YB-1 phosphorylation. Meanwhile, phosphorylation of YB-1 at S102, which is necessary for the majority of YB-1 functions, depends on the activity of ribosomal protein S6 kinase. LJI308 is a potent RSK inhibitor. The IC50s are 6 nM, 4 nM, and 13 nM for RSK1, RSK2, and RSK3, respectively.
 v
v
LJI308 is a specific and potent RSK inhibitor.
LJI308 is a potent RSK inhibitor. Moreover, it inhibits the phosphorylation of RSK (T359/S363) and YB-1 (S102) after irradiation, treatment with EGF, and in cells expressing a KRAS mutation. It also inhibits S6K1 with an IC50 of 0.8 μM. LJI308 also inhibits YB-1 phosphorylation in CRC cells at concentrations of 5 to 25 μM. In a dose kinetics experiment, LJI308, starting at 2.5 μM, inhibits YB-1 phosphorylation in the KRAS mutated TNBC cell line MDA-MB-231 by approximately 86%. In addition, LJI308 effectively blocks RSK and YB-1 phosphorylation after EGF stimulation and after irradiation in KRAS wild-type HBL-100 cells. Its treatment slightly inhibits DSB repair only in some of the cell lines.
In summary, inhibition of RSK by LJI308 is associated with activation of AKT. AKT activity also stimulates DSB repair and phosphorylates YB-1 as well. Thus, the cotargeting RSK and AKT may be an efficient approach to block DSB repair and improve radiation-induced clonogenic inactivation.
Reference:
Lettau K, et al. Int J Radiat Oncol Biol Phys. 2021;109(2):567-580.