Cdc42, a small GTPase of the Rho family, plays important roles in cytoskeleton organization, cell cycle progression, signal transduction, and vesicle trafficking. Overactive Cdc42 has been implicated in the pathology of cancers, immune diseases, and neuronal disorders. Therefore, Cdc42 inhibitors will be useful in probing molecular pathways and could have therapeutic potential. CID44216842 is a Cdc42 protein inhibitor. Moreover, CID44216842 demonstrates excellent selectivity with no inhibition toward Rho and Rac in the same GTPase family.
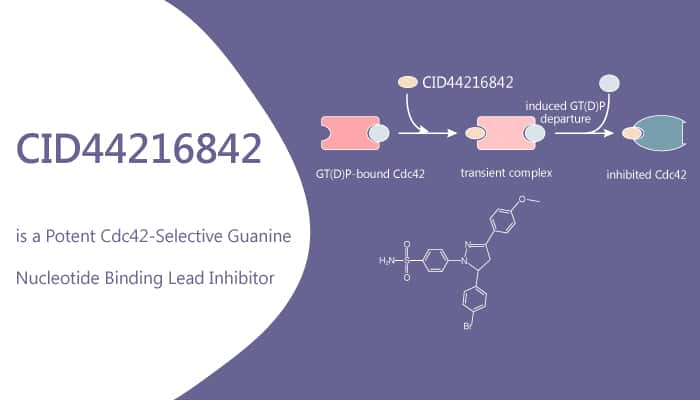
Biochemical characterization shows that CID44216842 acts as a non-competitive allosteric Cdc42 inhibitor. When tested in cellular assays, CID44216842 inhibits Cdc42-related filopodia formation and cell migration. Especially, CID44216842 significantly inhibits Cdc42 activation at 10 μM, whereas CID44216842 does not inhibit Rac1 or RhoA activation at the same concentration. CID44216842 inhibits stimulus-induced Cdc42 activation, but not Rac1 and RhoA. In addition, CID44216842 effects on RhoA activation are measured in Vero E6 kidney epithelial cells after Calpeptin stimulation. CID44216842 significantly inhibits Cdc42 activation at 10 μM, whereas CID44216842 does not inhibit Rac1 or RhoA activation at the same concentration.
In this study, researchers assess the cytotoxicity of CID44216842 in cells including neutrophils, fibroblasts, kidney epithelia, and ovarian cancer lines. Furthermore, CID44216842 is not cytotoxic toward U937ΔST cells at concentrations up to 10 μM for 24 h and up to 30 μM after 1 h.
All in all, CID44216842 is a potent Cdc42-selective guanine nucleotide-binding lead inhibitor. The EC50s for Cdc42 WT and Cdc42Q61L mutant are 1.0 and 1.2 μM in GTP binding assay, respectively. The EC50s for Cdc42 WT and Cdc42Q61L mutant are 0.3 and 0.5 μM in GDP binding assay, respectively.