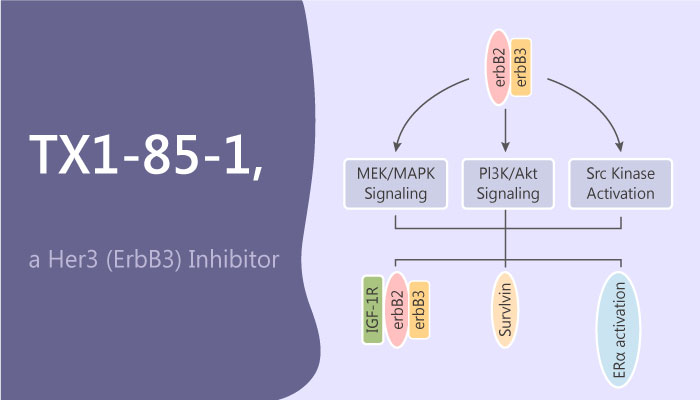
Her3 (also known as ErbB3) is a member of the epidermal growth factor receptor (EGFR) tyrosine kinases, including Her1, Her2 and Her4. However, owing to receptor or ligand overexpression or oncogenic mutations that results in constitutive activation of the kinase domain. EGFR family-dependent signaling frequently becomes deregulated in human cancers. On the one hand, it takes extensive efforts to treat patients with non-small-cell lung cancer (NSCLC) and breast cancer. Several FDA-approved drugs develop to target activated mutants of EGFR and Her2, such as Gefifitinib, Erlotinib, Lapatinib, and Afatinib. On the other hand, Her3 is well documented as an essential heterodimerization partner with EGFR, Her2, and c-Met.
In this study, author develops bifunctional molecules consisting of a covalent Her3 targeting small molecule, and it links to a hydrophobic adamantane moiety which induces partial Her3 degradation by the proteasome. Meanwhile, it simultaneously inhibits productive heterodimerization of Her3-Her2 and Her3–c-Met.
TX1-85-1 is a Her3 (ErbB3) inhibitor with an IC50 of 23 nM. It forms a covalent bond with Cys721 located in the ATP-binding site of Her3 to inhibit Her3 signaling.
Moreover, TX1-85-1 inhibits phosphorylation of Akt and ERK1/2 in ErbB3-dependent HCC2935 and OVCAR-8 cells when used at concentrations of 5 and 2 µM, respectively, and TX1-85-1 inhibits proliferation of ErbB3-dependent PC9 GR4, HCC827 GR8, or OVCAR-8 cells greater than or equal to approximately 10 μM (EC50s = ≥10 µM). In other words, TX1-85-1dose not effect proliferation in those Her3-dependent cancer cell lines. But, TX1-85-1 could induce partial degradation of Her3 protein and attenuates Her3-dependent signaling.
In summary, TX1-85-1 inhibits Her3 signaling by covalent modification of Her3. But, it dose not inhibit proliferation in some Her3-dependent cancer cell lines. Small molecules will be capable of perturbing the biological function of Her3 and ~60 other pseudokinases found in human cells.
Reference: