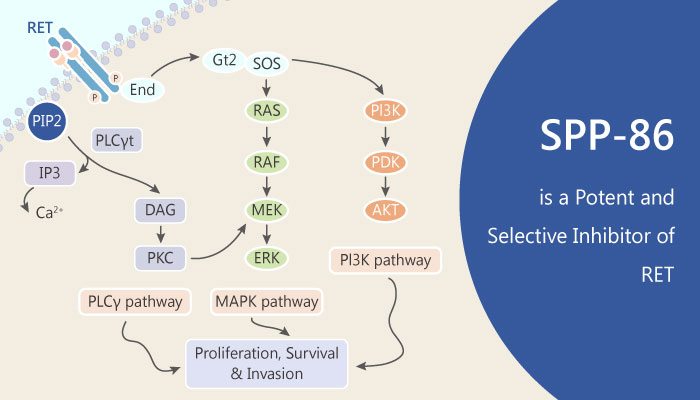
The REarranged during Transfection (RET) receptor tyrosine kinase (RTK) regulates key aspects of cellular proliferation and survival by regulating the activity of the mitogen- activated protein kinase (MAPK) and PI3K/Akt signaling pathways. The RET tyrosine kinase receptor has emerged as a target in thyroid and endocrine resistant breast cancer.
A study from John P Alao discovered and identified a RET kinase selective inhibitor, SPP-86. SPP-86 is a potent and selective cell permeable inhibitor of RET tyrosine kinase, with an IC50 of 8 nM.
In the study, the authors used several human cancer cells to carry out a series in vitro experiments.
Firstly, the authors found that SPP-86 effectively inhibits ERK1/2 phosphorylation in TPC1 cells expressing the RET/PTC1 rearrangement at a concentration of 1 μM. Inversely, the compound had no effect on ERK1/2 phosphorylation in 8505C or C643 cells.
Furthermore, in human breast cancer cell lines, estrogen deprived and serum starved MCF7 cells were exposed to 10 ng/mL GDNF in the absence or presence of increasing does of SPP-86. In these experiments, SPP-86 effectively inhibited GDNF/RET-induced ERα phosphorylation at a concentration of 0.1 μM. SPP-86 with the concentration of 1-10 μM, reduced ERα phosphorylation even below baseline levels. In addition, exposure of MCF7 cells to SPP86 was associated with a moderate decrease in ERα levels in these experiments.
During the study, we can know that SPP86, a selective inhibitor towards RET on cancer cell proliferation. SPP86 is cell permeable, potently inhibits RET activity in vitro and in vivo, and exhibits a unique selectivity profile that differs from previously reported inhibitors with activity towards this kinase.