The myotonic dystrophy-related Cdc42-binding kinases MRCKα and MRCKβ contribute to the regulation of actin-myosin cytoskeleton organization and dynamics, acting in concert with the Rho-associated coiled-coil kinases ROCK1 and ROCK2. A study from Mathieu Unbekandt discovered and identified azaindole compounds BDP8900 and BDP9066 as potent and selective MRCK inhibitors.
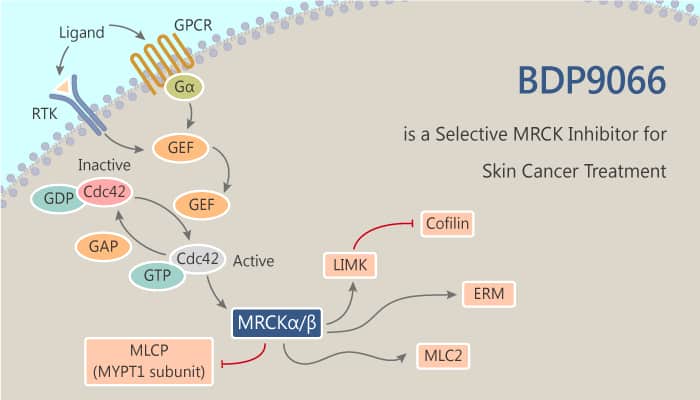
Both of the inhibitors could reduce substrate phosphorylation, leading to morphological changes in cancer cells along with inhibition of their motility and invasive character. BDP8900 and BDP9066 displayed consistent anti-proliferative effects with greatest activity in hematological cancer cells. And BDP9066 is today’s story.
In vitro, BDP9066 showed >100 times more selective for MRCKβ relative to ROCK1 or ROCK2. Under the assay conditions, BDP9066 was markedly selective against all kinases tested. In other words, BDP9066 are potent and highly selective inhibitors of MRCK with robust on-target actions in vitro and in whole cells. Besides, treatment of HEK293 cells expressing FLAG-tagged MRCKα, BDP90661 with the concentration of 1 µM blocked pS1003 immunoreactivity. Additionally, both autophosphorylation and MLC2 phosphorylation were blocked by BDP9066, supporting the relationship between MRCKα S1003 autophosphorylation and kinase activity. Furthermore, BDP9066 inhibits MLC phosphorylation and blocks SCC12 squamous cell carcinoma motility and invasion. And BDP9066 is relatively non-toxic at concentrations that profoundly inhibit substrate phosphorylation.
In vivo, Topical application of 25 µg BDP9066 led to 26 µM mean BDP9066 concentration in skin, but only 0.04 µM in blood. BDP9066 application led to significantly reduced epidermal MRCKα pS1003 positive staining. The results indicated that it was possible to achieve sustainable BDP9066 levels in mouse skin by repeated topical application. Moreover, BDP9066 was sufficient to induce phenotypic responses in squamous cell carcinoma cells in vitro, with low toxicity..