The BCL-2 family protein BAX is a central mediator of apoptosis. Besides, overexpression of anti-apoptotic BCL-2 proteins contributes to tumor development and resistance to therapy by suppressing BAX and its activators. Cancer cells depend on their ability to enforce cell survival pathways and block cell death mechanisms. Moreover, pro-apoptotic BAX is upon conformational activation and oligomerization at the mitochondrial outer membrane (MOM). Furthermore, it causes the permeabilization of the MOM and the release of mitochondrial factors. Cells deficient for BAX become less sensitive to various apoptotic stimuli. Moreover, the induction of BAX-dependent apoptosis by BH3-only proteins can be induced by several classic chemotherapeutic agents. Interestingly, the vast majority of cancer cells contain functional BAX in an inactive conformation or suppressed by anti-apoptotic proteins. BTSA1 is a potent, high affinity and orally active BAX activator with anti-cancer activity.
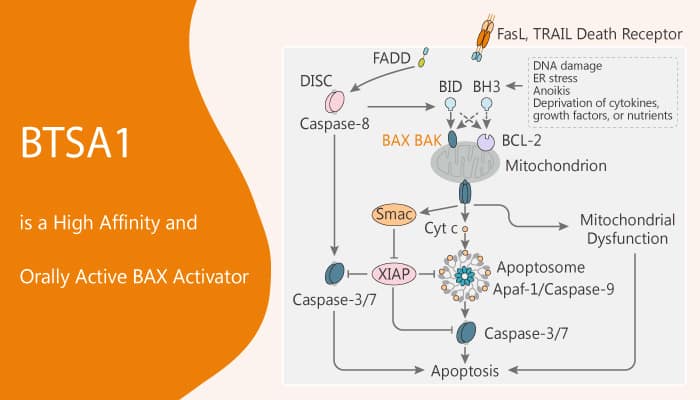
BTSA1 is a potent, high affinity and orally active BAX activator with an IC50 of 250 nM and an EC50 of 144 nM. In addition, BTSA1 binds with high affinity and specificity to the N-terminal activation site and induces conformational changes to BAX leading to BAX-mediated apoptosis. Meanwhile, BTSA1-induced BAX activation effectively promotes apoptosis in leukemia cell lines and patient samples while sparing healthy cells. Nonetheless, BAX expression levels and cytosolic conformation regulate sensitivity to BTSA1. Specifically, BTSA1 potently suppressed human acute myeloid leukemia (AML) xenografts and increased host survival without toxicity. All in all, BTSA1 is a potent, high affinity and orally active BAX activator with anti-cancer activity.
References:
Reyna DE, et al. Cancer Cell. 2017 Oct 9;32(4):490-505.e10.