Rho-GTPases are molecular switches that cycle between an inactive GDP-bound form and an active GTP-bound form. Guanine nucleotide exchange factors (GEFs) catalyze nucleotide exchange and mediate activation in this cycle, While GTPase-activating proteins (GAPs) stimulate GTP hydrolysis and inactivate the GTPase. Only the active GTP-bound state binds to downstream effector proteins and actively transduces signals. Rac1 is one of the members of the Rho-GTPases family. It controls fundamental cellular processes including cell proliferation, actin cytoskeleton reorganization, migration, cell cycle progression, cell adhesion, differentiation, and apoptosis. Rho GTPases play a key role in different cancer types. Thus, they are attractive and validated targets for anticancer therapies. Rac1 GTPase acts as a critical mediator of signaling pathways contributing to the interactions of hematopoietic stem cells with their microenvironment. In this study, ZINC69391, a specific Rac1 inhibitor, interferes with Rac1-GEF interaction by masking Trp56 residue on Rac1 surface.
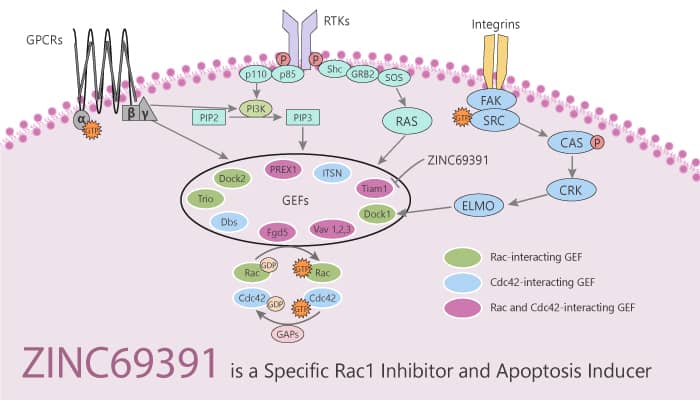
ZINC69391, a specific Rac1 inhibitor, interferes with Rac1-GEF interaction, induces apoptosis, shows antiproliferative, and antimetastatic effects.
ZINC69391 is a Rac1-GEF interaction inhibitor. It inhibits several Rac1-GEF interactions, which were associated with antiproliferative effects, cell cycle arrest, and migration inhibition of highly aggressive breast cancer cell lines. In addition, ZINC69391 inhibits the growth of U937, HL-60, KG1A, and Jurkat cells with IC50s ranging from 41 to 54 μM. Moreover, It augments the enzymatic activity of caspase 3 in a concentration-dependent manner. It also reduces cell proliferation of human glioma cells and triggers cell cycle arrest. In addition, ZINC69391 has in vivo anti-metastatic activity in lung and apoptotic induction in glioma cells with decreased cell migration and invasion. Furthermore, ZINC69391 also impairs metastatic lung colonization in a syngeneic animal model.
In summary, ZINC69391 is a specific Rac1 inhibitor. It interferes with Rac1-GEF interaction by masking Trp56 residue on Rac1 surface, and it induces apoptosis and shows antiproliferative and antimetastatic effects. The Rac1 pathway is a potential therapeutic target in hematopoietic malignancies.
Reference:
Cabrera M, et al. Oncotarget. 2017;8(58):98509‐98523. Published 2017 Oct 4.