Hypoxia is a non-physiological level of oxygen tension, a phenomenon common in a majority of malignant tumors. Hypoxia associates with tumor progression, resistance to therapy, and metastasis in cancer. Numerous pathways play critical roles in cancer maintenance, including noncoding RNAs such as microRNA (miR)-210 that regulates hypoxia-inducible factors. In this study, researchers describe the identification of a small molecule Targapremir-210. In particular, Targapremir-210 binds to the Dicer site of the miR-210 hairpin precursor.
Targapremir-210 selectively recognizes the miR-210 precursor and can differentially recognize RNAs in cells that have the same target motif but have different expression levels. Targapremir-210 selectively targets RNAs and affects cellular responses to environmental conditions, resulting in favorable benefits against cancer. Moreover, Targapremir-210 inhibits pre-miR-210 processing at nanomolar concentrations. Importantly, Targapremir-210 binds similarly to an in vitro transcribed pre-miR-210 and binds avidly to pre-miR-210 and binding sufficiently to inhibit its processing in vitro. Targapremir-210 decreases mature miR-210 levels in MDA-MB-231 cells cultured under hypoxic conditions with an IC50 of ∼200 nM. Indeed, treatment reduces HIF-1α levels and induces apoptosis.
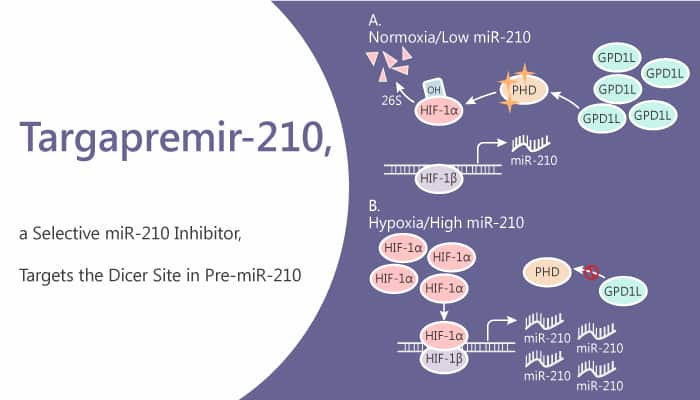
Interestingly, Targapremir-210 (200 nM) only affects the levels of mature miR-210, indicating that it is at least as selective as the oligonucleotide-based antagomir. Further, Targapremir-210 inhibits tumorigenesis in a mouse xenograft model of hypoxic triple-negative breast cancer. Furthermore, it decreases tumor burden in the triple-negative breast cancer (TNBC) mouse model and impedes tumor proliferation in vivo.
To summarise, Targapremir-210 binds miR-210’s Dicer processing site and modulates the miR-210 hypoxic circuit in triple-negative breast cancer cells and a mouse xenograft model.