Ras is a small GTPase and interacts with the protein Raf to create activated Ras-Raf complex that activates the downstream effectors in the ERK pathway. Malfunctioning Ras-Raf switches cause cancer. Ras-Raf interaction has taken a center stage in cancer research. Ras is a peripheral membrane protein that acts as a switch by binding to GDP or GTP. In particular, MCP110 is an inhibitor of Ras/Raf-1 interaction and blocks the interaction of Ras with Raf.
MCP110 reduces serum-induced transcriptional activation of serum response element as well as Ras-induced transcription by way of the AP-1 site. MCP110 also inhibits Ras-induced Raf-1 activation in human embryonic kidney 293 cells, Raf-1 and MAP2K1 activities in HT1080 fibrosarcoma cells, and EGF-induced Raf-1 activation in A549 lung carcinoma cells. In addition, MCP110 causes reversion of ras-transformed phenotypes including morphology, in vitro invasiveness, and anchorage-independent growth of HT1080 cells. Moreover, MCP110 also decreases level of matrix metalloproteinases.
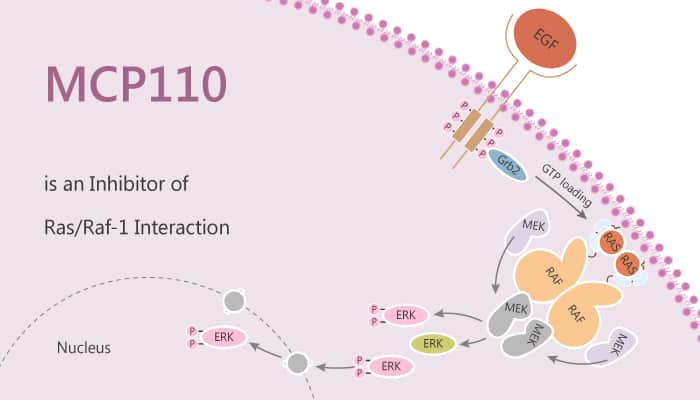
MCP110 restores actin stress fibers and causes flat reversion in NIH 3T3 cells transformed with H-Ras (V12) but not in NIH 3T3 cells transformed with constitutively active Raf-1 (RafΔN). Furthermore, MCP110 causes G1 enrichment of A549 cells with the decrease of cyclin D level. Besides, MCP110 also inhibits H-Ras-induced AP-1 activation in HEK293 cells. MCP110 shows a dose-dependent inhibition of this elevated Raf-1 activity. Furthermore, MCP110 decreases cyclin D level stimulated by the treatment with EGF, platelet-derived growth factor, serum, or phorbol 12-myristate 13-acetate. Finally, MCP110 inhibits anchorage-independent growth of A549 and PANC-1 cells harboring K-ras mutation.
Taken together, these results highlight potent and specific effects of MCP compounds on cancer cells with intrinsic Ras activation.