Focal adhesion kinase (FAK) is a tyrosine kinase overexpressed in cancer cells. Especially, FAK plays an important role in the progression of tumors to a malignant phenotype. FAK associates with integrins and modulates various cellular processes including growth, survival, and migration. In particular, PND-1186 is a substituted pyridine reversible inhibitor of FAK activity with an IC50 of 1.5 nM in vitro.
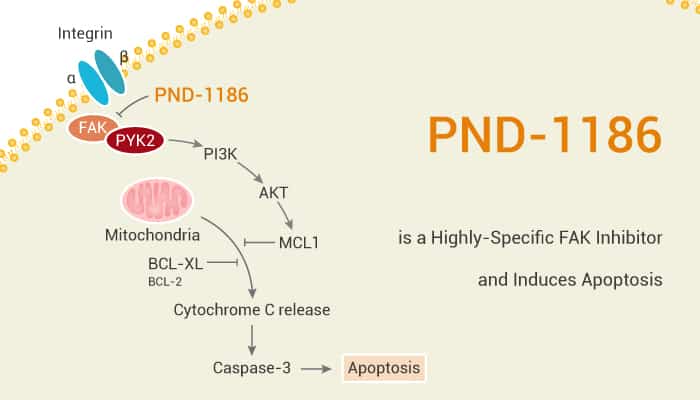
In the tumor microenvironment, nuclear FAK can regulate the formation of new blood vessels, affecting the tumor blood supply. PND-1186 has an IC50 of ~100 nM in breast carcinoma cells as determined by anti-phospho-specific immunoblotting to FAK Tyr-397. Moreover, PND-1186 blocks FAK and p130Cas tyrosine phosphorylation promotes caspase-3 activation and triggers cell apoptosis. PND-1186 inhibits 4T1 breast carcinoma subcutaneous tumor growth correlated with elevated tumor cell apoptosis and caspase 3 activations. Particularly, PND-1186 has an IC50 of 1.5 nM to recombinant FAK and ~0.1 µM in breast carcinoma cells as determined by anti-phospho-specific immunoblotting to FAK Tyr-397. Furthermore, PND-1186 inhibits breast carcinoma cell motility in a dose-dependent fashion.
PND-1186 inhibits 4T1 subcutaneous tumor growth and is associated with tumor cell apoptosis. In addition, PND-1186 functions as a potent preventative and/or prophylactic anti-tumor agent. Increasing concentrations of PND-1186 (0.1 to 1.0 µM) added to 4T1 cells inhibit FAK Tyr-397 phosphorylation (pY397) and result in elevated levels of total FAK protein within 1 h. Besides, PND-1186 inhibits 4T1 spheroid growth and FAK-p130Cas phosphorylation in suspension.
Taken together, FAK can affect the growth of cancer stem cells through this mechanism of the regulation of cell stiffness. Ultimately, PND-1186 potently inhibits FAK phosphorylation in a reversible manner.