The pseudokinase Mixed Lineage Kinase domain-like (MLKL) is an essential effector protein in the necroptotic cell death pathway downstream of the protein kinase Receptor Interacting Protein Kinase-3 (RIPK3). RIPK3–mediated phosphorylation of the activation loop in MLKL trips a molecular switch to induce necroptotic cell death. The structure of mouse MLKL revealing that MLKL contains a C-terminal pseudokinase domain and an N-terminal four-helix bundle (4HB) domain connected by a two-helix linker. The 4HB domain of MLKL is sufficient to induce necroptosis. It is able to oligomerize and translocate to membranes without an ectopic oligomerization domain or any other stimulus. In this study, GW806742X is an ATP mimetic and a potent MLKL (Mixed Lineage Kinase domain-like protein) inhibitor. GW806742X binds the MLKL pseudokinase domain with a Kd of 9.3 μM. It also retards MLKL membrane translocation and inhibits necroptosis. In addition, GW806742X has activity against VEGFR2 (IC50=2 nM).
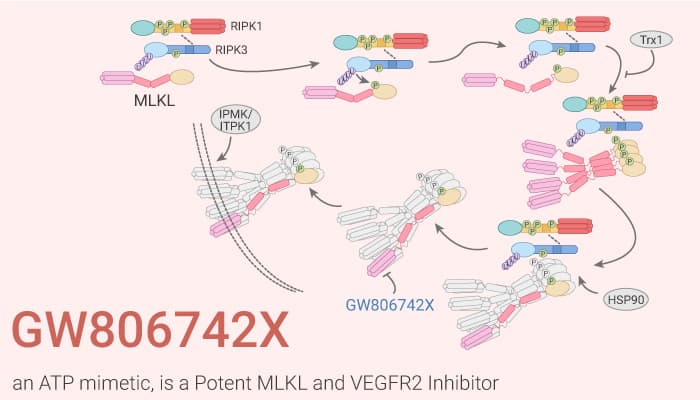
GW806742X bound the nucleotide-binding site in the MLKL pseudokinase domain because it competes with either ATP or ADP for binding to MLKL. GW806742X rescues 50% of wild-type MDFs from TSQ-induced necroptosis with an IC50 < 50 nM, with >50-fold greater potency than Nec-1, when 1 ng/mL TNF treatment. In addition, GW806742X shows inhibition of VEGF-induced proliferation of HUVECs with an IC50 of 5 nM.
GW806742X is a small molecule targeting the nucleotide-binding site of the MLKL pseudokinase domain. It also retards MLKL membrane translocation and inhibits necroptosis. Thus, GW806742X provides a novel tool to investigate necroptosis and demonstrates the feasibility of using small molecules to target the nucleotide binding site of pseudokinases to modulate signal transduction.
Reference:
Hildebrand JM, et al. Proc Natl Acad Sci U S A. 2014 Oct 21;111(42):15072-7.