GRP78 (Glucose-regulated protein, 78 kDa) is a key regulator of endoplasmic reticulum (ER) stress signaling. In this study, researchers describe the discovery of a series of GRP78 inhibitors. YUM70 is an ER stress inducer and kills cells by activating the apoptosis pathway. Especially, YUM70 treatment induces ER stress and triggers UPR by inhibiting GRP78.
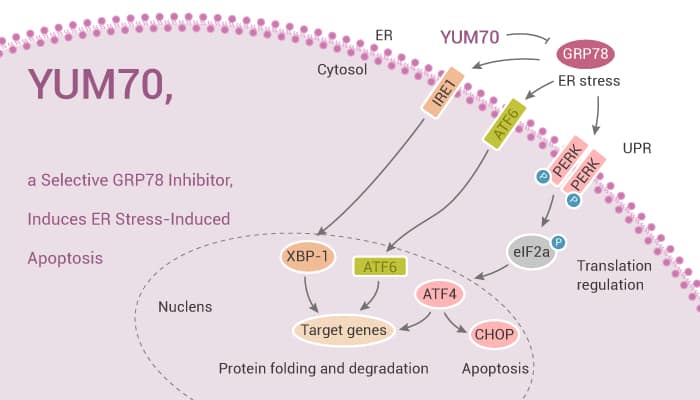
YUM70 inhibits pancreatic cancer cell growth in vitro and showed in vivo efficacy in a pancreatic cancer xenograft model with no toxicity to normal tissues. Importantly, YUM70 directly binds GRP78 and inactivates its function. Moreover, YUM70-PROTAC forces the degradation of GRP78 in pancreatic cancer cells.
YUM70 treatment upregulates ER stress-related genes, induces apoptosis, and demonstrates synergy with Topotecan and Vorinostat in killing pancreatic cancer cells. Interestingly, The IC50 of YUM70 varies among pancreatic cancer cell lines. IC50 of YUM70 in BxPC-3 is approximately three times higher than MIA PaCa-2 cells. PANC-1 and UM59 cells are also more sensitive to YUM70 than BxPC-3 cells. In addition, YUM70 induces ER stress-mediated apoptosis via phosphorylation of eIF2α. Treatment with YUM70 results in phosphorylation of eIF2α and increases in ATF4 and CHOP leading to apoptosis induction, and increased caspase 3/7 activity.
Mechanistically, YUM70 suppresses proliferation and induces ER stress and apoptosis in pancreatic cancer cells. YUM70 treatment significantly increases FAM129A protein levels.
In conclusion, GRP78 acts as a promising target to treat KRAS mutant pancreatic cancer and YUM70 as a potent anticancer agent that could be used in combination with select drugs to improve treatment efficacy and overcome drug resistance.